How auditory selectivity for sound timing arises: The diverse roles of GABAergic inhibition in shaping the excitation to interval-selective midbrain neurons
- PMID: 33242571
- PMCID: PMC7925425
- DOI: 10.1016/j.pneurobio.2020.101962
How auditory selectivity for sound timing arises: The diverse roles of GABAergic inhibition in shaping the excitation to interval-selective midbrain neurons
Abstract
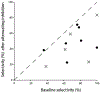
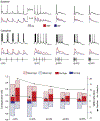
Across sensory systems, temporal frequency information is progressively transformed along ascending central pathways. Despite considerable effort to elucidate the mechanistic basis of these transformations, they remain poorly understood. Here we used a novel constellation of approaches, including whole-cell recordings and focal pharmacological manipulation, in vivo, and new computational algorithms that identify conductances resulting from excitation, inhibition and active membrane properties, to elucidate the mechanisms underlying the selectivity of midbrain auditory neurons for long temporal intervals. Surprisingly, we found that stimulus-driven excitation can be increased and its selectivity decreased following attenuation of inhibition with gabazine or intracellular delivery of fluoride. We propose that this nonlinear interaction is due to shunting inhibition. The rate-dependence of this inhibition results in the illusion that excitation to a cell shows greater temporal selectivity than is actually the case. We also show that rate-dependent depression of excitation, an important component of long-interval selectivity, can be decreased after attenuating inhibition. These novel findings indicate that nonlinear shunting inhibition plays a key role in shaping the amplitude and interval selectivity of excitation. Our findings provide a major advance in understanding how the brain decodes intervals and may explain paradoxical temporal selectivity of excitation to midbrain neurons reported previously.
Keywords: GABA; In vivo; Midbrain; Shunting inhibition; Temporal selectivity; Whole-cell recording.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Figures

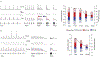
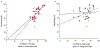



References
-
- ALDER TB & ROSE GJ 2000. Integration and recovery processes contribute to the temporal selectivity of neurons in the midbrain of the northern leopard frog, Rana pipiens. J Comp Physiol A, 186, 923–37. - PubMed
-
- ATKINS G, CHIBA A, ATKINS S & STOUT JF 1988. Low-pass filtering of sound signals by a high-frequency brain neuron and its input in the cricketAcheta domestica L. Journal of Comparative Physiology A, 164, 269–276.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

