Mirtazapine exerts astrocyte-mediated dopaminergic neuroprotection
- PMID: 33244123
- PMCID: PMC7693322
- DOI: 10.1038/s41598-020-77652-4
Mirtazapine exerts astrocyte-mediated dopaminergic neuroprotection
Abstract
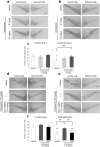
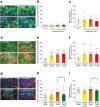
Mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA), is known to activate serotonin (5-HT) 1A receptor. Our recent study demonstrated that stimulation of astrocytic 5-HT1A receptors promoted astrocyte proliferation and upregulated antioxidative property in astrocytes to protect dopaminergic neurons against oxidative stress. Here, we evaluated the neuroprotective effects of mirtazapine against dopaminergic neurodegeneration in models of Parkinson's disease (PD). Mirtazapine administration attenuated the loss of dopaminergic neurons in the substantia nigra and increased the expression of the antioxidative molecule metallothionein (MT) in the striatal astrocytes of 6-hydroxydopamine (6-OHDA)-injected parkinsonian mice via 5-HT1A receptors. Mirtazapine protected dopaminergic neurons against 6-OHDA-induced neurotoxicity in mesencephalic neuron and striatal astrocyte cocultures, but not in enriched neuronal cultures. Mirtazapine-treated neuron-conditioned medium (Mir-NCM) induced astrocyte proliferation and upregulated MT expression via 5-HT1A receptors on astrocytes. Furthermore, treatment with medium from Mir-NCM-treated astrocytes protected dopaminergic neurons against 6-OHDA neurotoxicity, and these effects were attenuated by treatment with a MT-1/2-specific antibody or 5-HT1A antagonist. Our study suggests that mirtazapine could be an effective disease-modifying drug for PD and highlights that astrocytic 5-HT1A receptors may be a novel target for the treatment of PD.
Conflict of interest statement
The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

