An allosteric interleukin-1 receptor modulator mitigates inflammation and photoreceptor toxicity in a model of retinal degeneration
- PMID: 33246504
- PMCID: PMC7694438
- DOI: 10.1186/s12974-020-02032-8
An allosteric interleukin-1 receptor modulator mitigates inflammation and photoreceptor toxicity in a model of retinal degeneration
Abstract
Background: Inflammation and particularly interleukin-1β (IL-1β), a pro-inflammatory cytokine highly secreted by activated immune cells during early AMD pathological events, contribute significantly to retinal neurodegeneration. Here, we identify specific cell types that generate IL-1β and harbor the IL-1 receptor (IL-1R) and pharmacologically validate IL-1β's contribution to neuro-retinal degeneration using the IL-1R allosteric modulator composed of the amino acid sequence rytvela (as well as the orthosteric antagonist, Kineret) in a model of blue light-induced retinal degeneration.
Methods: Mice were exposed to blue light for 6 h and sacrificed 3 days later. Mice were intraperitoneally injected with rytvela, Kineret, or vehicle twice daily for 3 days. The inflammatory markers F4/80, NLRP3, caspase-1, and IL-1β were assessed in the retinas. Single-cell RNA sequencing was used to determine the cell-specific expression patterns of retinal Il1b and Il1r1. Macrophage-induced photoreceptor death was assessed ex vivo using retinal explants co-cultured with LPS-activated bone marrow-derived macrophages. Photoreceptor cell death was evaluated by the TUNEL assay. Retinal function was assessed by flash electroretinography.
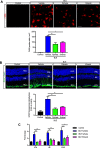
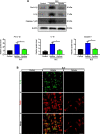
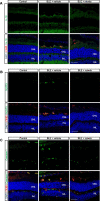
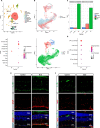
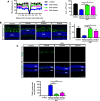
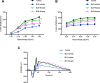
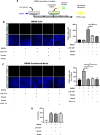
Results: Blue light markedly increased the mononuclear phagocyte recruitment and levels of inflammatory markers associated with photoreceptor death. Co-localization of NLRP3, caspase-1, and IL-1β with F4/80+ mononuclear phagocytes was clearly detected in the subretinal space, suggesting that these inflammatory cells are the main source of IL-1β. Single-cell RNA sequencing confirmed the immune-specific expression of Il1b and notably perivascular macrophages in light-challenged mice, while Il1r1 expression was found primarily in astrocytes, bipolar, and vascular cells. Retinal explants co-cultured with LPS/ATP-activated bone marrow-derived macrophages displayed a high number of TUNEL-positive photoreceptors, which was abrogated by rytvela treatment. IL-1R antagonism significantly mitigated the inflammatory response triggered in vivo by blue light exposure, and rytvela was superior to Kineret in preserving photoreceptor density and retinal function.
Conclusion: These findings substantiate the importance of IL-1β in neuro-retinal degeneration and revealed specific sources of Il1b from perivascular MPs, with its receptor Ilr1 being separately expressed on surrounding neuro-vascular and astroglial cells. They also validate the efficacy of rytvela-induced IL-1R modulation in suppressing detrimental inflammatory responses and preserving photoreceptor density and function in these conditions, reinforcing the rationale for clinical translation.
Keywords: Inflammasome; Inflammation; Interleukin-1; Interleukin-1 receptor; Photoreceptors; Retinal degeneration; Rytvela; cell death.
Conflict of interest statement
The corresponding author (SC) holds a patent on a small peptide antagonist of IL-1R, which could be useful in inflammatory ischemic retinopathies, yet this compound remains at the pre-clinical stage and the intellectual property is held by the CHU-Sainte Justine Hospital, Montreal.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

