Ribosome-associated quality control of membrane proteins at the endoplasmic reticulum
- PMID: 33247003
- PMCID: PMC7116877
- DOI: 10.1242/jcs.251983
Ribosome-associated quality control of membrane proteins at the endoplasmic reticulum
Abstract
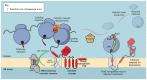
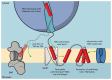
Protein synthesis is an energetically costly, complex and risky process. Aberrant protein biogenesis can result in cellular toxicity and disease, with membrane-embedded proteins being particularly challenging for the cell. In order to protect the cell from consequences of defects in membrane proteins, quality control systems act to maintain protein homeostasis. The majority of these pathways act post-translationally; however, recent evidence reveals that membrane proteins are also subject to co-translational quality control during their synthesis in the endoplasmic reticulum (ER). This newly identified quality control pathway employs components of the cytosolic ribosome-associated quality control (RQC) machinery but differs from canonical RQC in that it responds to biogenesis state of the substrate rather than mRNA aberrations. This ER-associated RQC (ER-RQC) is sensitive to membrane protein misfolding and malfunctions in the ER insertion machinery. In this Review, we discuss the advantages of co-translational quality control of membrane proteins, as well as potential mechanisms of substrate recognition and degradation. Finally, we discuss some outstanding questions concerning future studies of ER-RQC of membrane proteins.
Keywords: Endoplasmic reticulum; Membrane protein; Protein folding; Ribosome; Translation.
© 2020. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

