A Unified Framework for Dopamine Signals across Timescales
- PMID: 33248024
- PMCID: PMC7736562
- DOI: 10.1016/j.cell.2020.11.013
A Unified Framework for Dopamine Signals across Timescales
Abstract
Rapid phasic activity of midbrain dopamine neurons is thought to signal reward prediction errors (RPEs), resembling temporal difference errors used in machine learning. However, recent studies describing slowly increasing dopamine signals have instead proposed that they represent state values and arise independent from somatic spiking activity. Here we developed experimental paradigms using virtual reality that disambiguate RPEs from values. We examined dopamine circuit activity at various stages, including somatic spiking, calcium signals at somata and axons, and striatal dopamine concentrations. Our results demonstrate that ramping dopamine signals are consistent with RPEs rather than value, and this ramping is observed at all stages examined. Ramping dopamine signals can be driven by a dynamic stimulus that indicates a gradual approach to a reward. We provide a unified computational understanding of rapid phasic and slowly ramping dopamine signals: dopamine neurons perform a derivative-like computation over values on a moment-by-moment basis.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Interests The authors declare no competing interests.
Figures



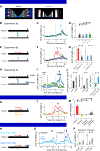

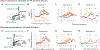

References
-
- Akaike H (1973). Information Theory as an Extension of the Maximum Likelihood Principle In Petrov BN and Csaki F (Eds.) Second International Symposium on Information Theory, (Budapest: Akadémiai Kiadó; ), pp. 267–281.
-
- Bäckman CM, Malik N, Zhang Y, Shan L, Grinberg A, Hoffer BJ, Westphal H, and Tomac AC (2006). Characterization of a mouse strain expressing Cre recombinase from the 3’ untranslated region of the dopamine transporter locus. Genesis 44, 383–390. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

