Mild heat stress changes the microbiota diversity in the respiratory tract and the cecum of layer-type pullets
- PMID: 33248618
- PMCID: PMC7704960
- DOI: 10.1016/j.psj.2020.09.024
Mild heat stress changes the microbiota diversity in the respiratory tract and the cecum of layer-type pullets
Abstract
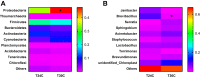
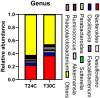
The present study aimed to research the effects of cyclic heat environment on the microbial diversity and structure of respiratory tract and cecum of chicken. A total of 360 layer-type pullets at 11 wk of age were subjected to different temperature treatments for 10 wk: constant 22°C; cyclic temperature 22°C to 24°C, 22°C to 26°C, 22°C to 28°C, 22°C to 30°C; the ambient temperature increased from 10:00, reached the set point within 1 h, and maintained until 18:00, thereafter the temperature was restored to 22°C; and the relative humidity was maintained at 60%. The result showed that feed intake of the chickens on ambient temperature 30°C group was significantly lower than that of the chickens on ambient temperature 24°C. The white blood cell, red blood cell, lymphocyte, hemoglobin, and pecked-cell volume content were highest at 24°C on 14, 16, and 18 wk. The ratio of CD3+CD4+/CD3+CD8+ T cells was lowest at 30°C. Meanwhile, the abundance of cecum bacteria in chickens at 30°C was lower than that at 24°C. Cyclic heat environment temperature treatment did not significantly affect the concentration of secretory immunoglobulin A in chicken bronchoalveolar lavage fluid (BALF) levels during 10 wk of trial. The diversity index analysis showed that the effect of 24°C on the cecum flora of chickens was optimal. Abundance of Firmicutes bacteria in the lung flora and cecum flora was lower at 30°C than at 24°C group. Similarly, the microorganism, Brevibacillus in the BALF was also significantly lower at 24°C. In conclusion, cyclic 24°C treatment was beneficial for the feed intake, blood routine indexes, microflora structure of the cecum, and respiratory tract in laying pullets.
Keywords: cecum microbiota; layer-type pullet; mild heat stress; respiratory microbiota.
Copyright © 2020. Published by Elsevier Inc.
Figures





Similar articles
-
Response of growth performance and cecum microbial community to cyclic heat stress in broilers.Trop Anim Health Prod. 2023 Dec 12;56(1):9. doi: 10.1007/s11250-023-03849-0. Trop Anim Health Prod. 2023. PMID: 38085433
-
Changes in the cecal microbiota of laying hens during heat stress is mainly associated with reduced feed intake.Poult Sci. 2019 Nov 1;98(11):5257-5264. doi: 10.3382/ps/pez440. Poult Sci. 2019. PMID: 31399742
-
The probiotic Bacillus licheniformis ameliorates heat stress-induced impairment of egg production, gut morphology, and intestinal mucosal immunity in laying hens.Poult Sci. 2012 Mar;91(3):575-82. doi: 10.3382/ps.2010-01293. Poult Sci. 2012. PMID: 22334732
-
Effects of two probiotic Lactobacillus strains on jejunal and cecal microbiota of broiler chicken under acute heat stress condition as revealed by molecular analysis of 16S rRNA genes.Microbiol Immunol. 2004;48(12):917-29. doi: 10.1111/j.1348-0421.2004.tb03620.x. Microbiol Immunol. 2004. PMID: 15611608
-
Quantitative review of the effects of environmental temperature on food intake, egg output and energy balance in laying pullets.Br Poult Sci. 1987 Dec;28(4):693-704. doi: 10.1080/00071668708417005. Br Poult Sci. 1987. PMID: 3328640 Review.
Cited by
-
Bacillus subtilis-Based Probiotic Improves Skeletal Health and Immunity in Broiler Chickens Exposed to Heat Stress.Animals (Basel). 2021 May 21;11(6):1494. doi: 10.3390/ani11061494. Animals (Basel). 2021. PMID: 34064126 Free PMC article. Review.
-
Effects of Different Ammonia Concentrations on Pulmonary Microbial Flora, Lung Tissue Mucosal Morphology, Inflammatory Cytokines, and Neurotransmitters of Broilers.Animals (Basel). 2022 Jan 21;12(3):261. doi: 10.3390/ani12030261. Animals (Basel). 2022. PMID: 35158583 Free PMC article.
-
Dietary supplementation of Ocimum gratissimum improves growth performance and immune response in broilers under high ambient temperature.J Anim Sci. 2023 Jan 3;101:skad212. doi: 10.1093/jas/skad212. J Anim Sci. 2023. PMID: 37348050 Free PMC article.
-
Yeast culture repairs rumen epithelial injury by regulating microbial communities and metabolites in sheep.Front Microbiol. 2023 Dec 1;14:1305772. doi: 10.3389/fmicb.2023.1305772. eCollection 2023. Front Microbiol. 2023. PMID: 38107864 Free PMC article.
-
Lack of consistency in poultry dust microbial taxa associated with high and low-performing commercial broiler flocks.Poult Sci. 2025 Jul;104(7):105173. doi: 10.1016/j.psj.2025.105173. Epub 2025 Apr 17. Poult Sci. 2025. PMID: 40267568 Free PMC article.
References
-
- Budden K.F., Gellatly S.L., Wood D.L.A., Cooper M.A., Morrison M., Hugenholtz P., Hansbro P.M. Emerging pathogenic links between microbiota and the gut–lung axis. Nat. Rev. Microbiol. 2017;15:55–63. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

