Switching to Progressively Reduced Nicotine Content Cigarettes in Smokers With Low Socioeconomic Status: A Double-Blind Randomized Clinical Trial
- PMID: 33249498
- PMCID: PMC8150128
- DOI: 10.1093/ntr/ntaa247
Switching to Progressively Reduced Nicotine Content Cigarettes in Smokers With Low Socioeconomic Status: A Double-Blind Randomized Clinical Trial
Abstract
Introduction: The Food and Drug Administration issued an advanced notice of proposed rulemaking for setting a product standard for nicotine levels in cigarettes, with an emphasis on minimally or non-addicting very low nicotine content (VLNC).
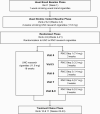
Methods: A 33 week, two-arm, double-blind randomized trial conducted in Hershey, Pennsylvania, USA and Washington, DC, USA included adult daily cigarette smokers (≥5 cigarettes per day) with less than a college degree, and who had no plans to quit within the next six months. Participants were randomized to either reduced nicotine content (RNC) study cigarettes tapered every three weeks to a final VLNC (0.2 mg/cigarette) for six weeks or to usual nicotine content (UNC) study cigarettes (11.6 mg/cigarette). Outcomes included acceptability of study cigarettes measured by attrition (primary outcome), compliance, reduction in cigarette dependence and tobacco biomarkers, and post-intervention cessation.
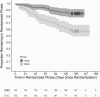
Results: The RNC (n = 122) versus UNC (n = 123) group had higher attrition (adjusted Hazard Ratio 3.4; 95% confidence interval [CI] 1.99 to 5.81). At the end of the intervention, cotinine levels were 50% lower in the RNC group (mean group difference -137 ng/mL; 95% CI -172, -102). The RNC group smoked fewer CPD (-4.1; 95% CI -6.44, -1.75) and had lower carbon monoxide levels (-4.0 ppm; 95% CI -7.7, -0.4). Forty seven percent (29/62) of the RNC group were biochemically-confirmed compliant with smoking VLNC cigarettes (mean cotinine = 8.9 ng/ml). At three month follow-up, only compliant VLNC smokers quit with an assisted quit attempt (N = 6/22, 27%).
Conclusions: This study supports a VLNC standard in cigarettes.
Implications: Differential dropout and noncompliance indicate some smokers had difficulty transitioning to cigarettes with reduced nicotine. These smokers will benefit from supplemental nicotine in medicinal or noncombustible tobacco products if a nicotine reduction standard is established. Other smokers successfully transitioned to very low nicotine content cigarettes exclusively and substantially reduced their exposure to nicotine.
© The Author(s) 2020. Published by Oxford University Press on behalf of the Society for Research on Nicotine and Tobacco. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

