Scale-Up Technologies for the Manufacture of Adherent Cells
- PMID: 33251241
- PMCID: PMC7672005
- DOI: 10.3389/fnut.2020.575146
Scale-Up Technologies for the Manufacture of Adherent Cells
Abstract
Great importance is being given to the impact our food supply chain and consumers' food habits are having on the environment, human health, and animal welfare. One of the latest developments aiming at positively changing the food ecosystem is represented by cultured meat. This form of cellular agriculture has the objective to generate slaughter-free meat products starting from the cultivation of few cells harvested from the animal tissue of interest. As a consequence, a large number of cells has to be generated at a reasonable cost. Just to give an idea of the scale, there were billions of cells just in a bite of the first cultured-meat burger. Thus, one of the major challenges faced by the scientists involved in this new ambitious and fascinating field, is how to efficiently scale-up cell manufacture. Considering the great potential presented by cultured meat, audiences from different backgrounds are very interested in this topic and eager to be informed of the challenges and possible solutions in this area. In light of this, we will provide an overview of the main existing bioprocessing technologies used to scale-up adherent cells at a small and large scale. Thus, giving a brief technical description of these bioprocesses, with the main associated advantages and disadvantages. Moreover, we will introduce an alternative solution we believe has the potential to revolutionize the way adherent cells are grown, helping cultured meat become a reality.
Keywords: adherent cell manufacture; bioprocessing; bioreactors; continuous bioprocessing; cultured meat; scale-up cells manufacture.
Copyright © 2020 Bellani, Ajeian, Duffy, Miotto, Groenewegen and Connon.
Figures
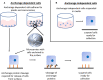

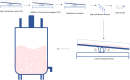
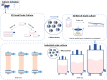

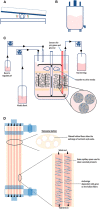

References
-
- Specht EA, Welch DR, Clayton EMR, Lagally CD. Opportunities for applying biomedical production and manufacturing methods to the development of the clean meat industry. Biochem Eng J. (2018) 132:161–8. 10.1016/j.bej.2018.01.015 - DOI
-
- Zhang G, Zhao X, Li X, Du G, Zhou J, Chen J. Challenges and possibilities for bio-manufacturing cultured meat. Trends Food Sci Technol. (2020) 97:443–50. 10.1016/j.tifs.2020.01.026 - DOI
-
- Lechanteur C, Baila S, Janssen ME, Giet O, Briquet A, Baudoux E, et al. Large-scale clinical expansion of mesenchymal stem cells in the GMP-compliant, closed automated quantum® cell expansion system: comparison with expansion in traditional T-flasks. J Stem Cell Res Therapy. (2014) 4:1000222 10.4172/2157-7633.1000222 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

