Flu Virus Attenuates Memory Clearance of Pneumococcus via IFN-γ-Dependent Th17 and Independent Antibody Mechanisms
- PMID: 33251497
- PMCID: PMC7683269
- DOI: 10.1016/j.isci.2020.101767
Flu Virus Attenuates Memory Clearance of Pneumococcus via IFN-γ-Dependent Th17 and Independent Antibody Mechanisms
Abstract
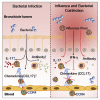
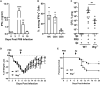
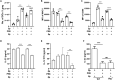
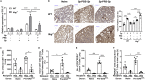
Bacterial coinfection is a major cause of influenza-associated mortality. Most people have experienced infections with bacterial pathogens commonly associated with influenza A virus (IAV) coinfection before IAV exposure; however, bacterial clearance through the immunological memory response (IMR) in coinfected patients is inefficient, suggesting that the IMR to bacteria is impaired during IAV infection. Adoptive transfer of CD4+ T cells from mice that had experienced bacterial infection into IAV-infected mice revealed that memory protection against bacteria was weakened in the latter. Additionally, memory Th17 cell responses were impaired due to an IFN-γ-dependent reduction in Th17 cell proliferation and delayed migration of CD4+ T cells into the lungs. A bacterium-specific antibody-mediated memory response was also substantially reduced in coinfected mice, independently of IFN-γ. These findings provide additional perspectives on the pathogenesis of coinfection and suggest additional strategies for the treatment of defective antibacterial immunity and the design of bacterial vaccines against coinfection.
Keywords: Immunology; Microbiology; Virology.
© 2020 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Memory Th17 cell-mediated protection against lethal secondary pneumococcal pneumonia following influenza infection.mBio. 2023 Aug 31;14(4):e0051923. doi: 10.1128/mbio.00519-23. Epub 2023 May 24. mBio. 2023. PMID: 37222516 Free PMC article.
-
Coinfection With Influenza A Virus and Klebsiella oxytoca: An Underrecognized Impact on Host Resistance and Tolerance to Pulmonary Infections.Front Immunol. 2018 Oct 29;9:2377. doi: 10.3389/fimmu.2018.02377. eCollection 2018. Front Immunol. 2018. PMID: 30420852 Free PMC article.
-
Influenza A virus infection impairs mycobacteria-specific T cell responses and mycobacterial clearance in the lung during pulmonary coinfection.J Immunol. 2013 Jul 1;191(1):302-11. doi: 10.4049/jimmunol.1202824. Epub 2013 May 22. J Immunol. 2013. PMID: 23698750
-
T cell mediated immunity to influenza: mechanisms of viral control.Trends Immunol. 2014 Aug;35(8):396-402. doi: 10.1016/j.it.2014.06.004. Epub 2014 Jul 16. Trends Immunol. 2014. PMID: 25043801 Review.
-
Modeling Influenza Virus Infection: A Roadmap for Influenza Research.Viruses. 2015 Oct 12;7(10):5274-304. doi: 10.3390/v7102875. Viruses. 2015. PMID: 26473911 Free PMC article. Review.
Cited by
-
Damping excessive viral-induced IFN-γ rescues the impaired anti-Aspergillus host immune response in influenza-associated pulmonary aspergillosis.EBioMedicine. 2024 Oct;108:105347. doi: 10.1016/j.ebiom.2024.105347. Epub 2024 Sep 30. EBioMedicine. 2024. PMID: 39353282 Free PMC article.
-
Mathematical Modeling of the Lethal Synergism of Coinfecting Pathogens in Respiratory Viral Infections: A Review.Microorganisms. 2023 Dec 13;11(12):2974. doi: 10.3390/microorganisms11122974. Microorganisms. 2023. PMID: 38138118 Free PMC article. Review.
-
Pathogenesis of Respiratory Viral and Fungal Coinfections.Clin Microbiol Rev. 2022 Jan 19;35(1):e0009421. doi: 10.1128/CMR.00094-21. Epub 2021 Nov 17. Clin Microbiol Rev. 2022. PMID: 34788127 Free PMC article. Review.
-
Memory Th17 cell-mediated protection against lethal secondary pneumococcal pneumonia following influenza infection.mBio. 2023 Aug 31;14(4):e0051923. doi: 10.1128/mbio.00519-23. Epub 2023 May 24. mBio. 2023. PMID: 37222516 Free PMC article.
References
-
- Beshara R., Sencio V., Soulard D., Barthelemy A., Fontaine J., Pinteau T., Deruyter L., Ismail M.B., Paget C., Sirard J.C. Alteration of Flt3-Ligand-dependent de novo generation of conventional dendritic cells during influenza infection contributes to respiratory bacterial superinfection. PLoS Pathog. 2018;14:e1007360. - PMC - PubMed
-
- Bromley S.K., Mempel T.R., Luster A.D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat. Immunol. 2008;9:970–980. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

