Prevalence of SARS-CoV-2 IgG antibodies in an area of northeastern Italy with a high incidence of COVID-19 cases: a population-based study
- PMID: 33253941
- PMCID: PMC7695553
- DOI: 10.1016/j.cmi.2020.11.013
Prevalence of SARS-CoV-2 IgG antibodies in an area of northeastern Italy with a high incidence of COVID-19 cases: a population-based study
Abstract
Objectives: A seroprevalence study of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was conducted in a high-incidence area located in northeastern Italy.
Methods: All citizens above 10 years of age resident in five municipalities of the Autonomous Province of Trento, with the highest incidence of coronavirus disease 2019 (COVID-19) cases, were invited to participate in the study. Among 6098 participants, 6075 sera and a standardized questionnaire administered face-to-face were collected between 5 May and 15 May 2020 and examined. Symptomatic individuals and their family contacts were tested by RT-PCR. Anti-SARS-CoV-2 antibodies were detected using an Abbott SARS-CoV-2 IgG assay, which was performed on the Abbott Architect i2000SR automated analyser. Seroprevalence was calculated as the proportion of positive results among the total number tested. A multivariable logistic regression model was performed to assess the relationship between seropositive versus seronegative individuals for a set of explanatory variables.
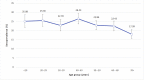
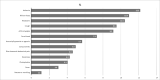
Results: A total of 1402 participants were positive for IgG antibodies against SARS-CoV-2, with a prevalence of 23.1% (1402/6075). The highest prevalence was found in the age class 40-49 years. Overall, 34.4% (2096/6098) of the participants reported at least one symptom. The ratio between reported cases identified by molecular test and those with seropositive results was 1:3, with a maximum ratio of about 1:7 in the age group <20 years and a minimum around 1:1 in those >70 years old. The infection fatality rate was 2.5% (35/1402). Among the symptoms, anosmia and ageusia were strongly associated with seropositivity.
Conclusions: The estimated seroprevalence of 23% was three-fold higher than the number of cases reported in the COVID-19 Integrated Surveillance data in the study area. This may be explained in part by a relatively high number of individuals presenting mild or no illness, especially those of younger age, and people who did not seek medical care or testing, but who may contribute to virus transmission in the community.
Keywords: IgG; Infection fatality rate; Population-based study; SARS-CoV-2; Seroprevalence.
Copyright © 2020 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

