Solid Lipid Nanoparticles as Formulative Strategy to Increase Oral Permeation of a Molecule Active in Multidrug-Resistant Tuberculosis Management
- PMID: 33255304
- PMCID: PMC7760137
- DOI: 10.3390/pharmaceutics12121132
Solid Lipid Nanoparticles as Formulative Strategy to Increase Oral Permeation of a Molecule Active in Multidrug-Resistant Tuberculosis Management
Abstract
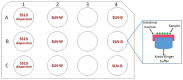
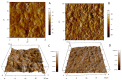
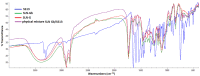
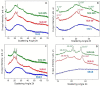
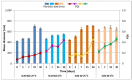
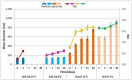
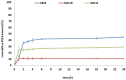
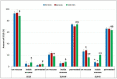
The role of mycobacterial efflux pumps in drug-resistant tuberculosis has been widely reported. Recently, a new compound, named SS13, has been synthesized, and its activity as a potential efflux inhibitor has been demonstrated. In this work, the chemical-physical properties of the SS13 were investigated; furthermore, a formulative study aimed to develop a formulation suitable for oral administration was performed. SS13 shows nonintrinsic antitubercular activity, but it increases the antitubercular activity of all the tested drugs on several strains. SS13 is insoluble in different simulated gastrointestinal media; thus, its oral absorption could be limited. Solid lipid nanoparticles (SLNs) were, therefore, developed by using two different lipids, Witepsol and/or Gelucire. Nanoparticles, having a particle size (range of 200-450 nm with regards to the formulation composition) suitable for intestinal absorption, are able to load SS13 and to improve its permeation through the intestinal mucosa compared to the pure compound. The cytotoxicity is influenced by the concentration of nanoparticles administered. These promising results support the potential application of these nanocarriers for increasing the oral permeation of SS13 in multidrug-resistant tuberculosis management.
Keywords: Gelucire@; Witepsol@; antitubercular activity; drug-resistant tuberculosis; oral permeation; solid lipid nanoparticles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Improving in vivo oral bioavailability of a poorly soluble drug: a case study on polymeric versus lipid nanoparticles.Drug Deliv Transl Res. 2023 Apr;13(4):1128-1139. doi: 10.1007/s13346-022-01278-4. Epub 2022 Dec 12. Drug Deliv Transl Res. 2023. PMID: 36509967
-
Formulation, optimization, and characterization of rifampicin-loaded solid lipid nanoparticles for the treatment of tuberculosis.Drug Dev Ind Pharm. 2018 Dec;44(12):1975-1989. doi: 10.1080/03639045.2018.1506472. Epub 2018 Aug 31. Drug Dev Ind Pharm. 2018. PMID: 30058392
-
Solid lipid nanoparticles for transdermal delivery of avanafil: optimization, formulation, in-vitro and ex-vivo studies.J Liposome Res. 2016 Dec;26(4):288-96. doi: 10.3109/08982104.2015.1117490. Epub 2016 Jan 19. J Liposome Res. 2016. PMID: 26784833
-
Use of Lipid Nanocarriers to Improve Oral Delivery of Vitamins.Nutrients. 2019 Jan 1;11(1):68. doi: 10.3390/nu11010068. Nutrients. 2019. PMID: 30609658 Free PMC article. Review.
-
[Development of antituberculous drugs: current status and future prospects].Kekkaku. 2006 Dec;81(12):753-74. Kekkaku. 2006. PMID: 17240921 Review. Japanese.
Cited by
-
Intestinal nanoparticle delivery and cellular response: a review of the bidirectional nanoparticle-cell interplay in mucosa based on physiochemical properties.J Nanobiotechnology. 2024 Nov 1;22(1):669. doi: 10.1186/s12951-024-02930-6. J Nanobiotechnology. 2024. PMID: 39487532 Free PMC article. Review.
-
Formulation design, production and characterisation of solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for the encapsulation of a model hydrophobic active.Food Hydrocoll Health. 2021;1:None. doi: 10.1016/j.fhfh.2021.100024. Food Hydrocoll Health. 2021. PMID: 35028634 Free PMC article.
-
Micro-nanoemulsion and nanoparticle-assisted drug delivery against drug-resistant tuberculosis: recent developments.Clin Microbiol Rev. 2023 Dec 20;36(4):e0008823. doi: 10.1128/cmr.00088-23. Epub 2023 Nov 30. Clin Microbiol Rev. 2023. PMID: 38032192 Free PMC article. Review.
-
Transmucosal Solid Lipid Nanoparticles to Improve Genistein Absorption via Intestinal Lymphatic Transport.Pharmaceutics. 2021 Feb 16;13(2):267. doi: 10.3390/pharmaceutics13020267. Pharmaceutics. 2021. PMID: 33669306 Free PMC article.
-
Surfactant-Free Chitosan/Cellulose Acetate Phthalate Nanoparticles: An Attempt to Solve the Needs of Captopril Administration in Paediatrics.Pharmaceuticals (Basel). 2022 May 25;15(6):662. doi: 10.3390/ph15060662. Pharmaceuticals (Basel). 2022. PMID: 35745581 Free PMC article.
References
-
- Taghizade Moghaddam H., Emami Moghadam Z., Khademi G., Bahreini A., Saeidi M. Tuberculosis: Past, present and future. Int. J. Pediatr. 2016;4:1243–1254.
-
- World Health Organization . Global Tuberculosis Report 2019. World Health Organization; Geneva, Switzerland: 2019.
-
- World Health Organization . WHO Guidelines on Tuberculosis Infection Prevention and Control: 2019 Update (No. WHO/CDS/TB/2019.1) World Health Organization; Geneva, Switzerland: 2019. - PubMed
-
- Zumla A., Chakaya J., Centis R., D’Ambrosio L., Mwaba P., Bates M., Kapata N., Nyirenda T., Chanda D., Mfinanga S., et al. Tuberculosis treatment and management-an update on treatment regimens, trials, new drugs, and adjunct therapies. Lancet Resp. Med. 2015;3:220–234. doi: 10.1016/S2213-2600(15)00063-6. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

