Adjuvanted Influenza Vaccines Elicits Higher Antibody Responses against the A(H3N2) Subtype than Non-Adjuvanted Vaccines
- PMID: 33255600
- PMCID: PMC7712667
- DOI: 10.3390/vaccines8040704
Adjuvanted Influenza Vaccines Elicits Higher Antibody Responses against the A(H3N2) Subtype than Non-Adjuvanted Vaccines
Abstract
Background: vaccination is the best approach to prevent influenza infections so far. Serological studies on the effect of different vaccine types are important to address vaccination campaigns and protect our population. In our study, we compared the serological response against influenza A subtypes using the non-adjuvanted influenza vaccine (NAIV) in adults and the elderly and the adjuvanted influenza vaccine (AIV) in the elderly.
Methods: We performed a retrospective analysis by hemagglutination inhibition assay (HI) of serum samples right before and 28 days after seasonal influenza vaccination during the 1996-2017 seasons.
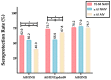
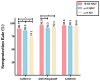
Conclusions: The AIV presents better performance against the A(H3N2) subtype in the elderly whereas the NAIV induces a better response against A(H1N1)pdm09 in the same group.
Keywords: Vaccines; adjuvanted Vaccines; influenza A.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Evaluation of non-inferiority of intradermal versus adjuvanted seasonal influenza vaccine using two serological techniques: a randomised comparative study.BMC Infect Dis. 2010 May 26;10:134. doi: 10.1186/1471-2334-10-134. BMC Infect Dis. 2010. PMID: 20504306 Free PMC article. Clinical Trial.
-
Non-neutralizing antibody responses following A(H1N1)pdm09 influenza vaccination with or without AS03 adjuvant system.Influenza Other Respir Viruses. 2021 Jan;15(1):110-120. doi: 10.1111/irv.12780. Epub 2020 Sep 5. Influenza Other Respir Viruses. 2021. PMID: 32889792 Free PMC article. Clinical Trial.
-
Immunogenicity of AS03-adjuvanted and non-adjuvanted trivalent inactivated influenza vaccines in elderly adults: A Phase 3, randomized trial and post-hoc correlate of protection analysis.Hum Vaccin Immunother. 2016 Dec;12(12):3043-3055. doi: 10.1080/21645515.2016.1219809. Hum Vaccin Immunother. 2016. PMID: 27690762 Free PMC article. Clinical Trial.
-
Priming with MF59 adjuvanted versus nonadjuvanted seasonal influenza vaccines in children - A systematic review and a meta-analysis.Vaccine. 2020 Jan 16;38(3):608-619. doi: 10.1016/j.vaccine.2019.10.053. Epub 2019 Nov 15. Vaccine. 2020. PMID: 31735505
-
Systematic review of the efficacy, effectiveness and safety of MF59® adjuvanted seasonal influenza vaccines for the prevention of laboratory-confirmed influenza in individuals ≥18 years of age.Rev Med Virol. 2023 May;33(3):e2329. doi: 10.1002/rmv.2329. Epub 2022 Feb 10. Rev Med Virol. 2023. PMID: 35142401
Cited by
-
Group 1 and group 2 hemagglutinin stalk antibody response according to age.Front Immunol. 2023 May 29;14:1194073. doi: 10.3389/fimmu.2023.1194073. eCollection 2023. Front Immunol. 2023. PMID: 37313413 Free PMC article.
References
-
- Iuliano A.D., Roguski K.M., Chang H.H., Muscatello D.J., Palekar R., Tempia S., Cohen C., Gran J.M., Schanzer D., Cowling B.J., et al. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet. 2018;391:1285–1300. doi: 10.1016/S0140-6736(17)33293-2. - DOI - PMC - PubMed
-
- Uyeki T.M., Bernstein H.H., Bradley J.S., Englund J.A., File T.M., Fry A.M., Gravenstein S., Hayden F.G., Harper S.A., Hirshon J.M., et al. Clinical Practice Guidelines by the Infectious Diseases Society of America: 2018 Update on Diagnosis, Treatment. Clin. Infect. Dis. 2019;68:1–99. doi: 10.1093/cid/ciy866. - DOI - PMC - PubMed
-
- Arriola C., Garg S., Anderson E.J., Ryan P.A., George A., Zansky S.M., Bennett N., Reingold A., Bargsten M., Miller L., et al. Influenza vaccination modifies disease severity among community-dwelling adults hospitalized with influenza. Clin. Infect. Dis. 2017;65:1289–1297. doi: 10.1093/cid/cix468. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

