Tetracyclic and Pentacyclic Triterpenes with High Therapeutic Efficiency in Wound Healing Approaches
- PMID: 33256207
- PMCID: PMC7730621
- DOI: 10.3390/molecules25235557
Tetracyclic and Pentacyclic Triterpenes with High Therapeutic Efficiency in Wound Healing Approaches
Abstract
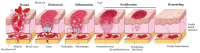
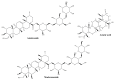
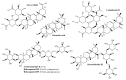
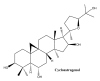
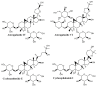
Wounds are among the most common skin conditions, displaying a large etiological diversity and being characterized by different degrees of severity. Wound healing is a complex process that involves multiple steps such as inflammation, proliferation and maturation and ends with scar formation. Since ancient times, a widely used option for treating skin wounds are plant- based treatments which currently have become the subject of modern pharmaceutical formulations. Triterpenes with tetracyclic and pentacyclic structure are extensively studied for their implication in wound healing as well as to determine their molecular mechanisms of action. The current review aims to summarize the main results of in vitro, in vivo and clinical studies conducted on lupane, ursane, oleanane, dammarane, lanostane and cycloartane type triterpenes as potential wound healing treatments.
Keywords: pentacyclic triterpenes; tetracyclic triterpenes; wound healing.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

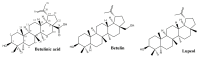

References
-
- Clark R.A.F. Wound repair: Basic biology to tissue engineering. In: Vacanti R., Lanza R., Langer J., editors. Principles of Tissue Engineering. 4th ed. Academic Press; London, UK: 2014. pp. 1595–1617.
-
- Nather A. The Diabetic Foot. World Scientific Pub. Co. Pte. Lt.; Singapore: 2012. pp. 89–96.
-
- Enoch S., Leaper D.J. Basic science of wound healing. Surgery (Oxford) 2008;26:31–37. doi: 10.1016/j.mpsur.2007.11.005. - DOI
-
- Prakash G., Varma S., Malhotra P. Prevention and Management of Infections. In: Sharma A., editor. Textbook of Systemic Vasculitis. Jaypee Brothers Medical Publishers; New Delhi, India: 2015. pp. 407–412.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

