Potential implications of DMET ontogeny on the disposition of commonly prescribed drugs in neonatal and pediatric intensive care units
- PMID: 33256492
- PMCID: PMC8346204
- DOI: 10.1080/17425255.2021.1858051
Potential implications of DMET ontogeny on the disposition of commonly prescribed drugs in neonatal and pediatric intensive care units
Abstract
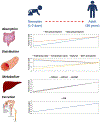
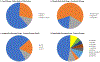
Introduction: Pediatric patients, especially neonates and infants, are more susceptible to adverse drug events as compared to adults. In particular, immature small molecule drug metabolism and excretion can result in higher incidences of pediatric toxicity than adults if the pediatric dose is not adjusted.Area covered: We reviewed the top 29 small molecule drugs prescribed in neonatal and pediatric intensive care units and compiled the mechanisms of their metabolism and excretion. The ontogeny of Phase I and II drug metabolizing enzymes and transporters (DMETs), particularly relevant to these drugs, are summarized. The potential effects of DMET ontogeny on the metabolism and excretion of the top pediatric drugs were predicted. The current regulatory requirements and recommendations regarding safe and effective use of drugs in children are discussed. A few representative examples of the use of ontogeny-informed physiologically based pharmacokinetic (PBPK) models are highlighted.Expert opinion: Empirical prediction of pediatric drug dosing based on body weight or body-surface area from the adult parameters can be inaccurate because DMETs are not mature in children and the age-dependent maturation of these proteins is different. Ontogeny-informed-PBPK modeling provides a better alternative to predict the pharmacokinetics of drugs in children.
Keywords: Pediatric precision medicine; developmental toxicity; drug metabolism; drug transport; ontogeny; pediatric pharmacology.
Conflict of interest statement
Declaration of interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Figures
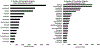

Similar articles
-
Developmental Expression of Drug Transporters and Conjugating Enzymes Involved in Enterohepatic Recycling: Implication for Pediatric Drug Dosing.Clin Pharmacol Ther. 2024 Dec;116(6):1615-1626. doi: 10.1002/cpt.3409. Epub 2024 Aug 19. Clin Pharmacol Ther. 2024. PMID: 39160670
-
Incorporating Ontogeny in Physiologically Based Pharmacokinetic Modeling to Improve Pediatric Drug Development: What We Know About Developmental Changes in Membrane Transporters.J Clin Pharmacol. 2019 Sep;59 Suppl 1(Suppl 1):S56-S69. doi: 10.1002/jcph.1489. J Clin Pharmacol. 2019. PMID: 31502692 Free PMC article. Review.
-
Predictive Pediatric Modeling and Simulation Using Ontogeny Information.J Clin Pharmacol. 2019 Sep;59 Suppl 1:S95-S103. doi: 10.1002/jcph.1497. J Clin Pharmacol. 2019. PMID: 31502689 Review.
-
Ontogeny of Hepatic Drug Transporters and Relevance to Drugs Used in Pediatrics.Drug Metab Dispos. 2016 Jul;44(7):992-8. doi: 10.1124/dmd.115.067801. Epub 2015 Dec 28. Drug Metab Dispos. 2016. PMID: 26712821 Review.
-
A repository of protein abundance data of drug metabolizing enzymes and transporters for applications in physiologically based pharmacokinetic (PBPK) modelling and simulation.Sci Rep. 2019 Jul 4;9(1):9709. doi: 10.1038/s41598-019-45778-9. Sci Rep. 2019. PMID: 31273226 Free PMC article.
Cited by
-
PBPK Modeling of Acetaminophen in Pediatric Populations: Incorporation of SULT Enzyme Ontogeny to Predict Age-Dependent Metabolism and Systemic Exposure.Life (Basel). 2025 Jul 13;15(7):1099. doi: 10.3390/life15071099. Life (Basel). 2025. PMID: 40724601 Free PMC article.
-
Developmental Expression of Drug Transporters and Conjugating Enzymes Involved in Enterohepatic Recycling: Implication for Pediatric Drug Dosing.Clin Pharmacol Ther. 2024 Dec;116(6):1615-1626. doi: 10.1002/cpt.3409. Epub 2024 Aug 19. Clin Pharmacol Ther. 2024. PMID: 39160670
-
Pediatric Drug Development: Reviewing Challenges and Opportunities by Tracking Innovative Therapies.Pharmaceutics. 2023 Oct 6;15(10):2431. doi: 10.3390/pharmaceutics15102431. Pharmaceutics. 2023. PMID: 37896191 Free PMC article. Review.
References
-
- Implementation of the Food Quality Protection Act. Food Drug Law J [Internet]. 1997;52:525. Available from: https://heinonline.org/HOL/Page?handle=hein.journals/foodlj52&id=563&div.... - PubMed
-
- Johnson TN, Rostami-Hodjegan A, Tucker GT. Prediction of the clearance of eleven drugs and associated variability in neonates, infants and children. Clin Pharmacokinet. 2006;45:931–956. - PubMed
-
- Sutherland JM. Fatal Cardiovascular Collapse of Infants Receiving Large Amounts of Chloramphenicol. AMA J Dis Child. 1959;97:761–767. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
