Audiological and otologic manifestations of glutaric aciduria type I
- PMID: 33256818
- PMCID: PMC7706203
- DOI: 10.1186/s13023-020-01571-w
Audiological and otologic manifestations of glutaric aciduria type I
Abstract
Background: Glutaric aciduria type 1 (GA-1) is a rare disease connected with speech delay and neurological deficits. However, the audiological and otologic profiles of GA-1 have not yet been fully characterized. To our knowledge, this is the largest study of comprehensive audiological and otologic evaluation in patients with GA-1 to date.
Methods: Thirteen patients diagnosed with GA-1 between January 1994 and December 2019 with audiological, radiological and genetic manifestations were retrospectively analyzed. Hearing tests were performed in all patients. MRI was performed for radiological evaluation.
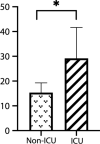
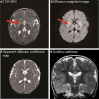
Results: Hearing loss was found in 76.9% (10/13) of GA-1 patients, including slight hearing loss in 46.1% (6/13) of patients, mild hearing loss in 15.4% (2/13) of patients, and moderate hearing loss in 7.7% (1/13) of patients. Normal hearing thresholds were seen in 23% (3/13) of patients. Patients with intensive care unit (ICU) admission history showed significantly worse hearing than those without (29.17 ± 12.47 vs 13.56 ± 3.93 dB HL, 95% CI 2.92-24.70, p = 0.0176). One patient had moderate sensorineural hearing loss and a past history of acute encephalopathic crisis. No usual causative gene mutations associated with hearing loss were found in these patients. MRI showed a normal vestibulocochlear apparatus and cochlear nerve. One patient with extensive injury of the basal ganglia on MRI after acute encephalopathic crisis was found to have moderate sensorineural hearing loss. Two patients with disability scores above 5 were found to have mild to moderate hearing impairment. No obvious correlation between macrocephaly and hearing loss was found.
Conclusion: A high prevalence of hearing impairment is found in GA-1 patients. Adequate audiological evaluation is essential for these patients, especially for those after encephalopathic crises or with ICU admission history.
Keywords: Glutaric aciduria type 1; Hereditary hearing loss; Syndromic hearing loss.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Does glutaric aciduria type 1 affect hearing function?Metab Brain Dis. 2022 Aug;37(6):2121-2132. doi: 10.1007/s11011-022-00987-6. Epub 2022 Apr 30. Metab Brain Dis. 2022. PMID: 35488943
-
A Korean patient with glutaric aciduria type 1 with a novel mutation in the glutaryl CoA dehydrogenase gene.Ann Clin Lab Sci. 2014 Spring;44(2):213-6. Ann Clin Lab Sci. 2014. PMID: 24795062
-
COVID-19 triggered encephalopathic crisis in a patient with glutaric aciduria type 1.J Pediatr Endocrinol Metab. 2021 Sep 14;34(12):1611-1614. doi: 10.1515/jpem-2021-0474. Print 2021 Dec 20. J Pediatr Endocrinol Metab. 2021. PMID: 34517439
-
[Complex heterogeneity phenotypes and genotypes of glutaric aciduria type 1].Zhongguo Dang Dai Er Ke Za Zhi. 2016 May;18(5):460-5. doi: 10.7499/j.issn.1008-8830.2016.05.016. Zhongguo Dang Dai Er Ke Za Zhi. 2016. PMID: 27165598 Free PMC article. Review. Chinese.
-
Recurrent rhabdomyolysis and glutaric aciduria type I: a case report and literature review.World J Pediatr. 2016 Aug;12(3):368-371. doi: 10.1007/s12519-016-0042-x. Epub 2016 Jun 29. World J Pediatr. 2016. PMID: 27351573 Review.
Cited by
-
The biochemical subtype is a predictor for cognitive function in glutaric aciduria type 1: a national prospective follow-up study.Sci Rep. 2021 Sep 29;11(1):19300. doi: 10.1038/s41598-021-98809-9. Sci Rep. 2021. PMID: 34588557 Free PMC article.
-
Sensorineural Hearing Loss in a Child with Succinic Semialdehyde Dehydrogenase Deficiency.Balkan J Med Genet. 2023 Jul 31;26(1):63-68. doi: 10.2478/bjmg-2023-0008. eCollection 2023 Jul. Balkan J Med Genet. 2023. PMID: 37576789 Free PMC article.
References
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

