Potential Drug Interactions of Repurposed COVID-19 Drugs with Lung Cancer Pharmacotherapies
- PMID: 33257051
- PMCID: PMC7670900
- DOI: 10.1016/j.arcmed.2020.11.006
Potential Drug Interactions of Repurposed COVID-19 Drugs with Lung Cancer Pharmacotherapies
Abstract
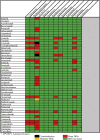
Lung cancer patients are at heightened risk for developing COVID-19 infection as well as complications due to multiple risk factors such as underlying malignancy, anti-cancer treatment induced immunosuppression, additional comorbidities and history of smoking. Recent literatures have reported a significant proportion of lung cancer patients coinfected with COVID-19. Chloroquine, hydroxychloroquine, lopinavir/ritonavir, ribavirin, oseltamivir, remdesivir, favipiravir, and umifenovir represent the major repurposed drugs used as potential experimental agents for COVID-19 whereas azithromycin, dexamethasone, tocilizumab, sarilumab, famotidine and ceftriaxone are some of the supporting agents that are under investigation for COVID-19 management. The rationale of this review is to identify potential drug-drug interactions (DDIs) occurring in lung cancer patients receiving lung cancer medications and repurposed COVID-19 drugs using Micromedex and additional literatures. This review has identified several potential DDIs that could occur with the concomitant treatments of COVID-19 repurposed drugs and lung cancer medications. This information may be utilized by the healthcare professionals for screening and identifying potential DDIs with adverse outcomes, based on their severity and documentation levels and consequently design prophylactic and management strategies for their prevention. Identification, reporting and management of DDIs and dissemination of related information should be a major consideration in the delivery of lung cancer care during this ongoing COVID-19 pandemic for better patient outcomes and updating guidelines for safer prescribing practices in this coinfected condition.
Keywords: COVID-19; Chemotherapy; Drug-drug interactions; Lung cancer; QT prolongation; Tyrosine kinase inhibitors.
Copyright © 2020 IMSS. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Repurposed pharmacological agents for the potential treatment of COVID-19: a literature review.Respir Res. 2021 Nov 27;22(1):304. doi: 10.1186/s12931-021-01885-8. Respir Res. 2021. PMID: 34838020 Free PMC article. Review.
-
Risk Assessment of Drug-Induced Long QT Syndrome for Some COVID-19 Repurposed Drugs.Clin Transl Sci. 2021 Jan;14(1):20-28. doi: 10.1111/cts.12882. Epub 2020 Nov 18. Clin Transl Sci. 2021. PMID: 32888379 Free PMC article. Review.
-
Drug-drug interactions with candidate medications used for COVID-19 treatment: An overview.Pharmacol Res Perspect. 2021 Feb;9(1):e00705. doi: 10.1002/prp2.705. Pharmacol Res Perspect. 2021. PMID: 33421347 Free PMC article. Review.
-
Drug-drug Interactions between COVID-19 Treatments and Antidepressants, Mood Stabilizers/Anticonvulsants, and Benzodiazepines: Integrated Evidence from 3 Databases.Pharmacopsychiatry. 2022 Jan;55(1):40-47. doi: 10.1055/a-1492-3293. Epub 2021 Jun 25. Pharmacopsychiatry. 2022. PMID: 34171927
-
Safety and efficacy of antiviral combination therapy in symptomatic patients of Covid-19 infection - a randomised controlled trial (SEV-COVID Trial): A structured summary of a study protocol for a randomized controlled trial.Trials. 2020 Oct 20;21(1):866. doi: 10.1186/s13063-020-04774-5. Trials. 2020. PMID: 33081849 Free PMC article. Clinical Trial.
Cited by
-
Scoping insight on antiviral drugs against COVID-19.Arab J Chem. 2021 Oct;14(10):103385. doi: 10.1016/j.arabjc.2021.103385. Epub 2021 Aug 16. Arab J Chem. 2021. PMID: 34909060 Free PMC article. Review.
-
Prediction of potential drug interactions between repurposed COVID-19 and antitubercular drugs: an integrational approach of drug information software and computational techniques data.Ther Adv Drug Saf. 2021 Aug 26;12:20420986211041277. doi: 10.1177/20420986211041277. eCollection 2021. Ther Adv Drug Saf. 2021. PMID: 34471515 Free PMC article.
-
Drug-Drug Interactions among Patients Hospitalized with COVID-19 in Greece.J Clin Med. 2022 Dec 2;11(23):7172. doi: 10.3390/jcm11237172. J Clin Med. 2022. PMID: 36498745 Free PMC article.
-
Exploration of the Potential Link, Hub Genes, and Potential Drugs for Coronavirus Disease 2019 and Lung Cancer Based on Bioinformatics Analysis.J Oncol. 2022 Sep 26;2022:8124673. doi: 10.1155/2022/8124673. eCollection 2022. J Oncol. 2022. PMID: 36199786 Free PMC article.
-
Computational methods directed towards drug repurposing for COVID-19: advantages and limitations.RSC Adv. 2021 Nov 10;11(57):36181-36198. doi: 10.1039/d1ra05320e. eCollection 2021 Nov 4. RSC Adv. 2021. PMID: 35492747 Free PMC article. Review.
References
-
- Kamboj M., Sepkowitz K.A. Nosocomial infections in patients with cancer. Lancet Oncol. 2009;10:589–597. - PubMed
-
- Longbottom E.R., Torrance H.D., Owen H.C. Features of postoperative immune suppression are reversible with interferon gamma and independent of interleukin-6 pathways. Ann Surg. 2016;264:370–377. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical