A Genetic Mouse Model Recapitulates Immune Checkpoint Inhibitor-Associated Myocarditis and Supports a Mechanism-Based Therapeutic Intervention
- PMID: 33257470
- PMCID: PMC8041233
- DOI: 10.1158/2159-8290.CD-20-0856
A Genetic Mouse Model Recapitulates Immune Checkpoint Inhibitor-Associated Myocarditis and Supports a Mechanism-Based Therapeutic Intervention
Abstract
Immune checkpoint inhibitors (ICI) targeting CTLA4 or PD-1/PD-L1 have transformed cancer therapy but are associated with immune-related adverse events, including myocarditis. Here, we report a robust preclinical mouse model of ICI-associated myocarditis in which monoallelic loss of Ctla4 in the context of complete genetic absence of Pdcd1 leads to premature death in approximately half of mice. Premature death results from myocardial infiltration by T cells and macrophages and severe ECG abnormalities, closely recapitulating the clinical and pathologic hallmarks of ICI-associated myocarditis observed in patients. Using this model, we show that Ctla4 and Pdcd1 functionally interact in a gene dosage-dependent manner, providing a mechanism by which myocarditis arises with increased frequency in the setting of combination ICI therapy. We demonstrate that intervention with CTLA4-Ig (abatacept) is sufficient to ameliorate disease progression and additionally provide a case series of patients in which abatacept mitigates the fulminant course of ICI myocarditis. SIGNIFICANCE: We provide a preclinical model of ICI-associated myocarditis which recapitulates this clinical syndrome. Using this model, we demonstrate that CTLA4 and PD-1 (ICI targets) functionally interact for myocarditis development and that intervention with CTLA4-Ig (abatacept) attenuates myocarditis, providing mechanistic rationale and preclinical support for therapeutic clinical studies.See related commentary by Young and Bluestone, p. 537.This article is highlighted in the In This Issue feature, p. 521.
©2020 American Association for Cancer Research.
Figures
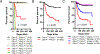


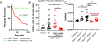
Comment in
-
At the Heart of Immune Checkpoint Inhibitor-Induced Immune Toxicity.Cancer Discov. 2021 Mar;11(3):537-539. doi: 10.1158/2159-8290.CD-21-0091. Cancer Discov. 2021. PMID: 33653918
References
-
- Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N Engl J Med [Internet]. 2015;373:23–34. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26027431 - PMC - PubMed
-
- Motzer RJ, Tannir NM, McDermott DF, Aren Frontera O, Melichar B, Choueiri TK, et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med [Internet]. 2018/03/22. 2018;378:1277–90. Available from: https://www.ncbi.nlm.nih.gov/pubmed/29562145 - PMC - PubMed
-
- Overman MJ, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, et al. Durable Clinical Benefit With Nivolumab Plus Ipilimumab in DNA Mismatch Repair-Deficient/Microsatellite Instability-High Metastatic Colorectal Cancer. J Clin Oncol [Internet]. 2018/01/23. 2018;36:773–9. Available from: https://www.ncbi.nlm.nih.gov/pubmed/29355075 - PubMed
-
- Postow MA, Chesney J, Pavlick AC, Robert C, Grossmann K, McDermott D, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med [Internet]. 2015;372:2006–17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25891304 - PMC - PubMed
-
- Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med [Internet]. 2013;369:122–33. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23724867 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 CA016672/CA/NCI NIH HHS/United States
- T32 GM007569/GM/NIGMS NIH HHS/United States
- R01 HL141466/HL/NHLBI NIH HHS/United States
- R35 HL144980/HL/NHLBI NIH HHS/United States
- R01 CA227481/CA/NCI NIH HHS/United States
- T32 GM007347/GM/NIGMS NIH HHS/United States
- P30 CA068485/CA/NCI NIH HHS/United States
- T32 GM152284/GM/NIGMS NIH HHS/United States
- R01 HL155990/HL/NHLBI NIH HHS/United States
- U24 DK059637/DK/NIDDK NIH HHS/United States
- S10 OD023475/OD/NIH HHS/United States
- R56 HL141466/HL/NHLBI NIH HHS/United States
- U2C DK059637/DK/NIDDK NIH HHS/United States
- F30 CA236157/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

