MRI-based anatomical characterisation of lower-limb muscles in older women
- PMID: 33259496
- PMCID: PMC7707470
- DOI: 10.1371/journal.pone.0242973
MRI-based anatomical characterisation of lower-limb muscles in older women
Abstract
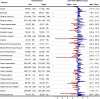
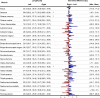
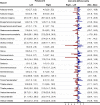
The ability of muscles to produce force depends, among others, on their anatomical features and it is altered by ageing-associated weakening. However, a clear characterisation of these features, highly relevant for older individuals, is still lacking. This study hence aimed at characterising muscle volume, length, and physiological cross-sectional area (PCSA) and their variability, between body sides and between individuals, in a group of post-menopausal women. Lower-limb magnetic resonance images were acquired from eleven participants (69 (7) y. o., 66.9 (7.7) kg, 159 (3) cm). Twenty-three muscles were manually segmented from the images and muscle volume, length and PCSA were calculated from this dataset. Personalised maximal isometric force was then calculated using the latter information. The percentage difference between the muscles of the two lower limbs was up to 89% and 22% for volume and length, respectively, and up to 84% for PCSA, with no recognisable pattern associated with limb dominance. Between-subject coefficients of variation reached 36% and 13% for muscle volume and length, respectively. Generally, muscle parameters were similar to previous literature, but volumes were smaller than those from in-vivo young adults and slightly higher than ex-vivo ones. Maximal isometric force was found to be on average smaller than those obtained from estimates based on linear scaling of ex-vivo-based literature values. In conclusion, this study quantified for the first time anatomical asymmetry of lower-limb muscles in older women, suggesting that symmetry should not be assumed in this population. Furthermore, we showed that a scaling approach, widely used in musculoskeletal modelling, leads to an overestimation of the maximal isometric force for most muscles. This heavily questions the validity of this approach for older populations. As a solution, the unique dataset of muscle segmentation made available with this paper could support the development of alternative population-based scaling approaches, together with that of automatic tools for muscle segmentation.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
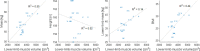

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

