Characterization of a Murine Model for Encephalitozoon hellem Infection after Dexamethasone Immunosuppression
- PMID: 33260440
- PMCID: PMC7761425
- DOI: 10.3390/microorganisms8121891
Characterization of a Murine Model for Encephalitozoon hellem Infection after Dexamethasone Immunosuppression
Abstract
Background: Encephalitozoon hellem (E. hellem) belongs to a group of opportunistic pathogens called microsporidia. Microsporidia infection symptoms vary and include diarrhea, ocular disorders and systemic inflammations. Traditionally, immunodeficient animals were used to study microsporidia infection. To overcome the difficulties in maintenance and operation using immunodeficient mice, and to better mimic natural occurring microsporidia infection, this study aims to develop a pharmacologically immunosuppressed murine model of E. hellem infection.
Methods: Wild-type C57BL/6 mice were immunosuppressed with dexamethasone (Dex) and then E. hellem spores were inoculated into the mice intraperitoneally. Control groups were the Dex-immunosuppressed but noninoculated mice, and the Dex-immunosuppressed then lipopolysaccharide (LPS)-treated mice. Mice body weights were monitored and all animals were sacrificed at the 15th day after inoculation. Tissue fragments and immune cells were collected and processed.
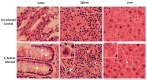
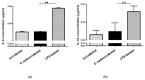
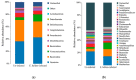
Results: Histopathological analysis demonstrated that E. hellem inoculation resulted in a disseminated nonlethal infection. Interestingly, E. hellem infection desensitized the innate immunity of the host, as shown by cytokine expressions and dendritic cell maturation. We also found that E. hellem infection greatly altered the composition of host gut microbiota. (4) Conclusions: Dex-immunosuppressed mice provide a useful tool for study microsporidiosis and the interactions between microsporidia and host immunity.
Keywords: Encephalitozoon hellem; dexamethasone; immunity; microsporidia; murine model.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Encephalitozoon hellem Infection Promotes Monocytes Extravasation.Pathogens. 2022 Aug 15;11(8):914. doi: 10.3390/pathogens11080914. Pathogens. 2022. PMID: 36015036 Free PMC article.
-
Experimental microsporidiosis in immunocompetent and immunodeficient mice and monkeys.Folia Parasitol (Praha). 1994;41(1):1-11. Folia Parasitol (Praha). 1994. PMID: 8050748
-
Von Willebrand Factor Facilitates Intravascular Dissemination of Microsporidia Encephalitozoon hellem.Front Cell Infect Microbiol. 2021 May 21;11:694957. doi: 10.3389/fcimb.2021.694957. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34095003 Free PMC article.
-
Therapeutic strategies for human microsporidia infections.Expert Rev Anti Infect Ther. 2005 Jun;3(3):419-34. doi: 10.1586/14787210.3.3.419. Expert Rev Anti Infect Ther. 2005. PMID: 15954858 Review.
-
In vitro cultivation of microsporidia of clinical importance.Clin Microbiol Rev. 2002 Jul;15(3):401-13. doi: 10.1128/CMR.15.3.401-413.2002. Clin Microbiol Rev. 2002. PMID: 12097248 Free PMC article. Review.
Cited by
-
E. hellem Ser/Thr protein phosphatase PP1 targets the DC MAPK pathway and impairs immune functions.Life Sci Alliance. 2024 Jan 10;7(4):e202302375. doi: 10.26508/lsa.202302375. Print 2024 Apr. Life Sci Alliance. 2024. PMID: 38199846 Free PMC article.
-
Huangqin Tea Total Flavonoids-Gut Microbiota Interactions: Based on Metabolome and Microbiome Analysis.Foods. 2023 Dec 7;12(24):4410. doi: 10.3390/foods12244410. Foods. 2023. PMID: 38137214 Free PMC article.
-
Peptide targeting improves the delivery and therapeutic index of glucocorticoids to treat rheumatoid arthritis.J Control Release. 2024 Apr;368:329-343. doi: 10.1016/j.jconrel.2024.02.040. Epub 2024 Mar 5. J Control Release. 2024. PMID: 38431094 Free PMC article.
-
Encephalitozoon hellem Infection Promotes Monocytes Extravasation.Pathogens. 2022 Aug 15;11(8):914. doi: 10.3390/pathogens11080914. Pathogens. 2022. PMID: 36015036 Free PMC article.
-
Microsporidia persistence in host impairs epithelial barriers and increases chances of inflammatory bowel disease.Microbiol Spectr. 2024 Feb 6;12(2):e0361023. doi: 10.1128/spectrum.03610-23. Epub 2023 Dec 27. Microbiol Spectr. 2024. PMID: 38149855 Free PMC article.
References
-
- del Aguila C., Lopez-Velez R., Fenoy S., Turrientes C., Cobo J., Navajas R., Visvesvara G.S., Croppo G.P., Da Silva A.J., Pieniazek N.J. Identification of Enterocytozoon bieneusi spores in respiratory samples from an AIDS patient with a 2-year history of intestinal microsporidiosis. J. Clin. Microbiol. 1997;35:1862–1866. doi: 10.1128/JCM.35.7.1862-1866.1997. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

