Chloroquine reverses chemoresistance via upregulation of p21WAF1/CIP1 and autophagy inhibition in ovarian cancer
- PMID: 33277461
- PMCID: PMC7718923
- DOI: 10.1038/s41419-020-03242-x
Chloroquine reverses chemoresistance via upregulation of p21WAF1/CIP1 and autophagy inhibition in ovarian cancer
Abstract
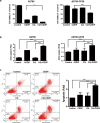
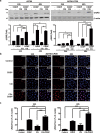
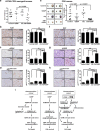
Overcoming drug-resistance is a big challenge to improve the survival of patients with epithelial ovarian cancer (EOC). In this study, we investigated the effect of chloroquine (CQ) and its combination with cisplatin (CDDP) in drug-resistant EOC cells. We used the three EOC cell lines CDDP-resistant A2780-CP20, RMG-1 cells, and CDDP-sensitive A2780 cells. The CQ-CDDP combination significantly decreased cell proliferation and increased apoptosis in all cell lines. The combination induced expression of γH2AX, a DNA damage marker protein, and induced G2/M cell cycle arrest. Although the CQ-CDDP combination decreased protein expression of ATM and ATR, phosphorylation of ATM was increased and expression of p21WAF1/CIP1 was also increased in CQ-CDDP-treated cells. Knockdown of p21WAF1/CIP1 by shRNA reduced the expression of γH2AX and phosphorylated ATM and inhibited caspase-3 activity but induced ATM protein expression. Knockdown of p21WAF1/CIP1 partly inhibited CQ-CDDP-induced G2/M arrest, demonstrating that knockdown of p21WAF1/CIP1 overcame the cytotoxic effect of the CQ-CDDP combination. Ectopic expression of p21WAF1/CIP1 in CDDP-treated ATG5-shRNA/A2780-CP20 cells increased expression of γH2AX and caspase-3 activity, demonstrating increased DNA damage and cell death. The inhibition of autophagy by ATG5-shRNA demonstrated similar results upon CDDP treatment, except p21WAF1/CIP1 expression. In an in vivo efficacy study, the CQ-CDDP combination significantly decreased tumor weight and increased expression of γH2AX and p21WAF1/CIP1 in A2780-CP20 orthotopic xenografts and a drug-resistant patient-derived xenograft model of EOC compared with controls. These results demonstrated that CQ increases cytotoxicity in combination with CDDP by inducing lethal DNA damage by induction of p21WAF1/CIP1 expression and autophagy inhibition in CDDP-resistant EOC.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

