Analysis of Long Noncoding RNA ZNF667-AS1 as a Potential Biomarker for Diagnosis and Prognosis of Glioma Patients
- PMID: 33282010
- PMCID: PMC7685845
- DOI: 10.1155/2020/8895968
Analysis of Long Noncoding RNA ZNF667-AS1 as a Potential Biomarker for Diagnosis and Prognosis of Glioma Patients
Abstract
Objective: Long noncoding RNAs (lncRNAs) have been strongly associated with various types of cancer. The present study aimed at exploring the diagnostic and prognostic value of lncRNA Zinc finger protein 667-antisense RNA 1 (ZNF667-AS1) in glioma patients. Patients and Methods. The expressions of ZNF667-AS1 were detected in 155 glioma tissues and matched normal brain tissue samples by qRT-PCR. The receiver operating characteristic (ROC) curve was performed to estimate the diagnostic value of ZNF667-AS1. The association between the ZNF667-AS1 expression and clinicopathological characteristics was analyzed by the chi-square test. The Kaplan-Meier method was performed to determine the influence of the ZNF667-AS1 expression on the overall survival and disease-free survival of glioma patients. The Cox regression analysis was used to evaluate the effect of independent prognostic factors on survival outcome. Cell proliferation was measured by the respective cell counting Kit-8 (CCK-8) assays.
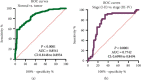
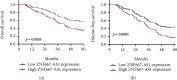
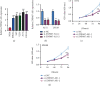
Results: We observed that ZNF667-AS1 was significantly upregulated in glioma tissues compared to normal tissue samples (p < 0.01). Higher levels of ZNF667-AS1 were positively associated with the WHO grade (p = 0.018) and KPS score (p = 0.008). ROC assays revealed that the high ZNF667-AS1 expression had an AUC value of 0.8541 (95% CI: 0.8148 to 0.8934) for glioma. Survival data revealed that glioma patients in the high ZNF667-AS1 expression group had significantly shorter 5-year overall survival (p = 0.0026) and disease-free survival (p = 0.0005) time than those in the low ZNF667-AS1 expression group. Moreover, multivariate analyses confirmed that the ZNF667-AS1 expression was an independent predictor of the overall survival and disease-free survival for glioma patients. Functionally, we found that knockdown of ZNF667-AS1 suppressed the proliferation of glioma cells.
Conclusions: Our results suggest that ZNF667-AS1 could be used as a potential diagnostic and prognostic biomarker in glioma.
Copyright © 2020 Qin Yuan et al.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

