Co-movement of astral microtubules, organelles and F-actin by dynein and actomyosin forces in frog egg cytoplasm
- PMID: 33284105
- PMCID: PMC7759381
- DOI: 10.7554/eLife.60047
Co-movement of astral microtubules, organelles and F-actin by dynein and actomyosin forces in frog egg cytoplasm
Abstract
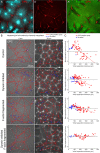
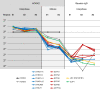
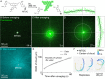
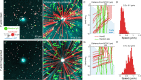
How bulk cytoplasm generates forces to separate post-anaphase microtubule (MT) asters in Xenopus laevis and other large eggs remains unclear. Previous models proposed that dynein-based, inward organelle transport generates length-dependent pulling forces that move centrosomes and MTs outwards, while other components of cytoplasm are static. We imaged aster movement by dynein and actomyosin forces in Xenopus egg extracts and observed outward co-movement of MTs, endoplasmic reticulum (ER), mitochondria, acidic organelles, F-actin, keratin, and soluble fluorescein. Organelles exhibited a burst of dynein-dependent inward movement at the growing aster periphery, then mostly halted inside the aster, while dynein-coated beads moved to the aster center at a constant rate, suggesting organelle movement is limited by brake proteins or other sources of drag. These observations call for new models in which all components of the cytoplasm comprise a mechanically integrated aster gel that moves collectively in response to dynein and actomyosin forces.
Keywords: ER; actin; cell biology; cytoplasm; dynein; mechanics; microtubule; physics of living systems; xenopus.
© 2020, Pelletier et al.
Conflict of interest statement
JP, CF, SF, MS, TM No competing interests declared
Figures












Similar articles
-
Microtubules remodel actomyosin networks in Xenopus egg extracts via two mechanisms of F-actin transport.J Cell Biol. 2000 Jul 24;150(2):361-76. doi: 10.1083/jcb.150.2.361. J Cell Biol. 2000. PMID: 10908578 Free PMC article.
-
Microtubule-dependent pushing forces contribute to long-distance aster movement and centration in Xenopus laevis egg extracts.Mol Biol Cell. 2020 Dec 1;31(25):2791-2802. doi: 10.1091/mbc.E20-01-0088. Epub 2020 Oct 7. Mol Biol Cell. 2020. PMID: 33026931 Free PMC article.
-
Microtubule aster formation by dynein-dependent organelle transport.Cell Motil Cytoskeleton. 1998;41(3):254-63. doi: 10.1002/(SICI)1097-0169(1998)41:3<254::AID-CM6>3.0.CO;2-4. Cell Motil Cytoskeleton. 1998. PMID: 9829779
-
Cytoplasmic microtubule-based motor proteins.Curr Opin Cell Biol. 1993 Feb;5(1):95-104. doi: 10.1016/s0955-0674(05)80014-6. Curr Opin Cell Biol. 1993. PMID: 8448036 Review.
-
Centering and Shifting of Centrosomes in Cells.Cells. 2020 May 29;9(6):1351. doi: 10.3390/cells9061351. Cells. 2020. PMID: 32485978 Free PMC article. Review.
Cited by
-
Spatial variation of microtubule depolymerization in large asters.Mol Biol Cell. 2021 Apr 19;32(9):869-879. doi: 10.1091/mbc.E20-11-0723. Epub 2021 Jan 13. Mol Biol Cell. 2021. PMID: 33439671 Free PMC article.
-
Cytoplasmic organization promotes protein diffusion in Xenopus extracts.Nat Commun. 2022 Sep 23;13(1):5599. doi: 10.1038/s41467-022-33339-0. Nat Commun. 2022. PMID: 36151204 Free PMC article.
-
Spring-like behavior of cytoplasm holds the mitotic spindle in place.Proc Natl Acad Sci U S A. 2022 Apr 5;119(14):e2203036119. doi: 10.1073/pnas.2203036119. Epub 2022 Mar 24. Proc Natl Acad Sci U S A. 2022. PMID: 35324318 Free PMC article. No abstract available.
-
Live-cell imaging under centrifugation characterized the cellular force for nuclear centration in the Caenorhabditis elegans embryo.bioRxiv [Preprint]. 2024 Jan 5:2024.01.03.574024. doi: 10.1101/2024.01.03.574024. bioRxiv. 2024. Update in: Proc Natl Acad Sci U S A. 2024 Oct 22;121(43):e2402759121. doi: 10.1073/pnas.2402759121. PMID: 38260704 Free PMC article. Updated. Preprint.
-
Contribution of cytoplasm viscoelastic properties to mitotic spindle positioning.Proc Natl Acad Sci U S A. 2022 Feb 22;119(8):e2115593119. doi: 10.1073/pnas.2115593119. Proc Natl Acad Sci U S A. 2022. PMID: 35169074 Free PMC article.
References
-
- Chambers R. Microdissection studies. II. The cell aster: a reversible gelation phenomenon. Journal of Experimental Zoology. 1917;23:483–505. doi: 10.1002/jez.1400230302. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

