Associations of Observational and Genetically Determined Caffeine Intake With Coronary Artery Disease and Diabetes Mellitus
- PMID: 33287642
- PMCID: PMC7955399
- DOI: 10.1161/JAHA.120.016808
Associations of Observational and Genetically Determined Caffeine Intake With Coronary Artery Disease and Diabetes Mellitus
Abstract
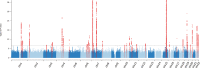
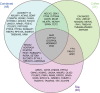
Background Caffeine is the most widely consumed psychostimulant and is associated with lower risk of coronary artery disease (CAD) and type 2 diabetes mellitus (T2DM). However, whether these associations are causal remains unknown. This study aimed to identify genetic variants associated with caffeine intake, and to investigate evidence for causal links with CAD or T2DM. In addition, we aimed to replicate previous observational findings. Methods and Results Observational associations were tested within UK Biobank using Cox regression analyses. Moderate observational caffeine intakes from coffee or tea were associated with lower risks of CAD or T2DM, with the lowest risks at intakes of 121 to 180 mg/day from coffee for CAD (hazard ratio [HR], 0.77 [95% CI, 0.73-0.82; P<1×10-16]), and 301 to 360 mg/day for T2DM (HR, 0.76 [95% CI, 0.67-0.86]; P=1.57×10-5). Next, genome-wide association studies were performed on self-reported caffeine intake from coffee, tea, or both in 407 072 UK Biobank participants. These analyses identified 51 novel genetic variants associated with caffeine intake at P<1.67×10-8. These loci were enriched for central nervous system genes. However, in contrast to the observational analyses, 2-sample Mendelian randomization analyses using the identified loci in independent disease-specific cohorts yielded no evidence for causal links between genetically determined caffeine intake and the development of CAD or T2DM. Conclusions Mendelian randomization analyses indicate genetically determined higher caffeine intake might not protect against CAD or T2DM, despite protective associations in observational analyses.
Keywords: Mendelian randomization; caffeine intake; coronary artery disease; genetics; type 2 diabetes mellitus.
Conflict of interest statement
None.
Figures


References
-
- Fitt E, Pell D, Cole D. Assessing caffeine intake in the United Kingdom diet. Food Chem. 2013;140:421–426. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

