Polyunsaturated fatty acid biosynthesis pathway determines ferroptosis sensitivity in gastric cancer
- PMID: 33288688
- PMCID: PMC7768719
- DOI: 10.1073/pnas.2006828117
Polyunsaturated fatty acid biosynthesis pathway determines ferroptosis sensitivity in gastric cancer
Abstract
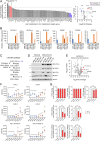
Ferroptosis is an iron-dependent regulated necrosis mediated by lipid peroxidation. Cancer cells survive under metabolic stress conditions by altering lipid metabolism, which may alter their sensitivity to ferroptosis. However, the association between lipid metabolism and ferroptosis is not completely understood. In this study, we found that the expression of elongation of very long-chain fatty acid protein 5 (ELOVL5) and fatty acid desaturase 1 (FADS1) is up-regulated in mesenchymal-type gastric cancer cells (GCs), leading to ferroptosis sensitization. In contrast, these enzymes are silenced by DNA methylation in intestinal-type GCs, rendering cells resistant to ferroptosis. Lipid profiling and isotope tracing analyses revealed that intestinal-type GCs are unable to generate arachidonic acid (AA) and adrenic acid (AdA) from linoleic acid. AA supplementation of intestinal-type GCs restores their sensitivity to ferroptosis. Based on these data, the polyunsaturated fatty acid (PUFA) biosynthesis pathway plays an essential role in ferroptosis; thus, this pathway potentially represents a marker for predicting the efficacy of ferroptosis-mediated cancer therapy.
Keywords: ELOVL5; FADS1; arachidonic acid; ferroptosis; lipid peroxidation.
Conflict of interest statement
Competing interest statement: Y.M.H. is a founder of Novomics Co., Ltd. and E.J., J.K., and Jihye Kim are full-time employees of Novomics Co., Ltd. The authors declare no competing interest.
Figures




References
-
- Seiler A., et al. , Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab. 8, 237–248 (2008). - PubMed
-
- Hassannia B., Vandenabeele P., Vanden Berghe T., Targeting ferroptosis to iron out cancer. Cancer Cell 35, 830–849 (2019). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

