Metarhizium: jack of all trades, master of many
- PMID: 33292103
- PMCID: PMC7776561
- DOI: 10.1098/rsob.200307
Metarhizium: jack of all trades, master of many
Abstract
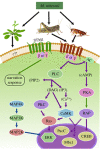
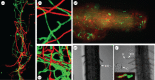
The genus Metarhizium and Pochonia chlamydosporia comprise a monophyletic clade of highly abundant globally distributed fungi that can transition between long-term beneficial associations with plants to transitory pathogenic associations with frequently encountered protozoans, nematodes or insects. Some very common 'specialist generalist' species are adapted to particular soil and plant ecologies, but can overpower a wide spectrum of insects with numerous enzymes and toxins that result from extensive gene duplications made possible by loss of meiosis and associated genome defence mechanisms. These species use parasexuality instead of sex to combine beneficial mutations from separate clonal individuals into one genome (Vicar of Bray dynamics). More weakly endophytic species which kill a narrow range of insects retain sexuality to facilitate host-pathogen coevolution (Red Queen dynamics). Metarhizium species can fit into numerous environments because they are very flexible at the genetic, physiological and ecological levels, providing tractable models to address how new mechanisms for econutritional heterogeneity, host switching and virulence are acquired and relate to diverse sexual life histories and speciation. Many new molecules and functions have been discovered that underpin Metarhizium associations, and have furthered our understanding of the crucial ecology of these fungi in multiple habitats.
Keywords: Metarhizium and Pochonia chlamydosporia; insect killing (entomopathogen); parasexual and asexual life histories (Red Queen and Vicar of Bray); parasitism to insects or nematodes; plant endophyte and symbiont; sexual; virulence evolution (host switching and speciation).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Lord JC. 2005. From Metchnikoff to Monsanto and beyond: the path of microbial control. J. Invert. Pathol. 89, 19–29. - PubMed
-
- Butt TM, Coates CJ, Dubovskiy IM, Ratcliffe NA. 2016. Entomopathogenic fungi: new insights into host–pathogen interactions. In Advances in genetics, vol. 94, pp. 307–364. New York, NY: Academic Press. - PubMed
-
- Lu HL, St. Leger RJ. 2016. Insect immunity to entomopathogenic fungi. In Advances in genetics, Vol. 94, pp. 251–285. Academic Press. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
