Walking the tight wire between cell adhesion and WNT signalling: a balancing act for β-catenin
- PMID: 33292105
- PMCID: PMC7776579
- DOI: 10.1098/rsob.200267
Walking the tight wire between cell adhesion and WNT signalling: a balancing act for β-catenin
Abstract
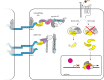
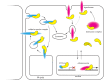
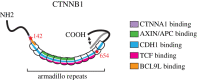
CTNNB1 (catenin β-1, also known as β-catenin) plays a dual role in the cell. It is the key effector of WNT/CTNNB1 signalling, acting as a transcriptional co-activator of TCF/LEF target genes. It is also crucial for cell adhesion and a critical component of cadherin-based adherens junctions. Two functional pools of CTNNB1, a transcriptionally active and an adhesive pool, can therefore be distinguished. Whether cells merely balance the distribution of available CTNNB1 between these functional pools or whether interplay occurs between them has long been studied and debated. While interplay has been indicated upon artificial modulation of cadherin expression levels and during epithelial-mesenchymal transition, it is unclear to what extent CTNNB1 exchange occurs under physiological conditions and in response to WNT stimulation. Here, we review the available evidence for both of these models, discuss how CTNNB1 binding to its many interaction partners is controlled and propose avenues for future studies.
Keywords: WNT signal transduction; cell adhesion; e-cadherin; epithelial–mesenchymal transition; interplay; β-catenin.
Conflict of interest statement
We declare we have no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
