Anakinra treatment in critically ill COVID-19 patients: a prospective cohort study
- PMID: 33302991
- PMCID: PMC7726611
- DOI: 10.1186/s13054-020-03364-w
Anakinra treatment in critically ill COVID-19 patients: a prospective cohort study
Abstract
Background: A subset of critically ill COVID-19 patients develop a hyperinflammatory state. Anakinra, a recombinant interleukin-1 receptor antagonist, is known to be effective in several hyperinflammatory diseases. We investigated the effects of anakinra on inflammatory parameters and clinical outcomes in critically ill, mechanically ventilated COVID-19 patients with clinical features of hyperinflammation.
Methods: In this prospective cohort study, 21 critically ill COVID-19 patients treated with anakinra were compared to a group of standard care. Serial data of clinical inflammatory parameters and concentrations of multiple circulating cytokines were determined and aligned on start day of anakinra in the treatment group, and median start day of anakinra in the control group. Analysis was performed for day - 10 to + 10 relative to alignment day. Clinical outcomes were analyzed during 28 days. Additionally, three sensitivity analyses were performed: (1) using propensity score-matched groups, (2) selecting patients who did not receive corticosteroids, and (3) using a subset of the control group aimed to match the criteria (fever, elevated ferritin) for starting anakinra treatment.
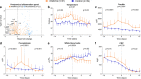
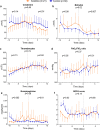
Results: Baseline patient characteristics and clinical parameters on ICU admission were similar between groups. As a consequence of bias by indication, plasma levels of aspartate aminotransferase (ASAT) (p = 0.0002), ferritin (p = 0.009), and temperature (p = 0.001) were significantly higher in the anakinra group on alignment day. Following treatment, no relevant differences in kinetics of circulating cytokines were observed between both groups. Decreases of clinical parameters, including temperature (p = 0.03), white blood cell counts (p = 0.02), and plasma levels of ferritin (p = 0.003), procalcitonin (p = 0.001), creatinine (p = 0.01), and bilirubin (p = 0.007), were more pronounced in the anakinra group. No differences in duration of mechanical ventilation or ICU length of stay were observed between groups. Sensitivity analyses confirmed these results.
Conclusions: Anakinra is effective in reducing clinical signs of hyperinflammation in critically ill COVID-19 patients. A randomized controlled trial is warranted to draw conclusion about the effects of anakinra on clinical outcomes.
Keywords: Coronavirus disease 2019; Critical care; Cytokines; Immunity; Interleukin-1 receptor antagonist protein; SARS-CoV-2.
Conflict of interest statement
The authors declare no conflict of interest or competing interest.
Figures


Comment in
-
Fast recovery of cardiac function in PIMS-TS patients early using intravenous anti-IL-1 treatment.Crit Care. 2021 Apr 7;25(1):131. doi: 10.1186/s13054-021-03548-y. Crit Care. 2021. PMID: 33827651 Free PMC article. No abstract available.
References
-
- Henry BM, de Oliveira MHS, Benoit S, Plebani M, Lippi G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 2020;58(7):1021–1028. doi: 10.1515/cclm-2020-0369. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

