Cadherin 11 Promotes Immunosuppression and Extracellular Matrix Deposition to Support Growth of Pancreatic Tumors and Resistance to Gemcitabine in Mice
- PMID: 33307028
- PMCID: PMC7956114
- DOI: 10.1053/j.gastro.2020.11.044
Cadherin 11 Promotes Immunosuppression and Extracellular Matrix Deposition to Support Growth of Pancreatic Tumors and Resistance to Gemcitabine in Mice
Abstract
Background & aims: Pancreatic ductal adenocarcinomas (PDACs) are characterized by fibrosis and an abundance of cancer-associated fibroblasts (CAFs). We investigated strategies to disrupt interactions among CAFs, the immune system, and cancer cells, focusing on adhesion molecule CDH11, which has been associated with other fibrotic disorders and is expressed by activated fibroblasts.
Methods: We compared levels of CDH11 messenger RNA in human pancreatitis and pancreatic cancer tissues and cells with normal pancreas, and measured levels of CDH11 protein in human and mouse pancreatic lesions and normal tissues. We crossed p48-Cre;LSL-KrasG12D/+;LSL-Trp53R172H/+ (KPC) mice with CDH11-knockout mice and measured survival times of offspring. Pancreata were collected and analyzed by histology, immunohistochemistry, and (single-cell) RNA sequencing; RNA and proteins were identified by imaging mass cytometry. Some mice were given injections of PD1 antibody or gemcitabine and survival was monitored. Pancreatic cancer cells from KPC mice were subcutaneously injected into Cdh11+/+ and Cdh11-/- mice and tumor growth was monitored. Pancreatic cancer cells (mT3) from KPC mice (C57BL/6), were subcutaneously injected into Cdh11+/+ (C57BL/6J) mice and mice were given injections of antibody against CDH11, gemcitabine, or small molecule inhibitor of CDH11 (SD133) and tumor growth was monitored.
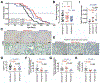
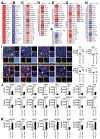
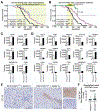
Results: Levels of CDH11 messenger RNA and protein were significantly higher in CAFs than in pancreatic cancer epithelial cells, human or mouse pancreatic cancer cell lines, or immune cells. KPC/Cdh11+/- and KPC/Cdh11-/- mice survived significantly longer than KPC/Cdh11+/+ mice. Markers of stromal activation entirely surrounded pancreatic intraepithelial neoplasias in KPC/Cdh11+/+ mice and incompletely in KPC/Cdh11+/- and KPC/Cdh11-/- mice, whose lesions also contained fewer FOXP3+ cells in the tumor center. Compared with pancreatic tumors in KPC/Cdh11+/+ mice, tumors of KPC/Cdh11+/- mice had increased markers of antigen processing and presentation; more lymphocytes and associated cytokines; decreased extracellular matrix components; and reductions in markers and cytokines associated with immunosuppression. Administration of the PD1 antibody did not prolong survival of KPC mice with 0, 1, or 2 alleles of Cdh11. Gemcitabine extended survival of KPC/Cdh11+/- and KPC/Cdh11-/- mice only or reduced subcutaneous tumor growth in mT3 engrafted Cdh11+/+ mice when given in combination with the CDH11 antibody. A small molecule inhibitor of CDH11 reduced growth of pre-established mT3 subcutaneous tumors only if T and B cells were present in mice.
Conclusions: Knockout or inhibition of CDH11, which is expressed by CAFs in the pancreatic tumor stroma, reduces growth of pancreatic tumors, increases their response to gemcitabine, and significantly extends survival of mice. CDH11 promotes immunosuppression and extracellular matrix deposition, and might be developed as a therapeutic target for pancreatic cancer.
Keywords: Activated Stroma; Anti-Tumor Immunity; Desmoplasia; Immunomodulation; Inflammation.
Copyright © 2021 AGA Institute. All rights reserved.
Conflict of interest statement
Disclosures:
S.W.B. and S.D. have formed a company Diviner Therapeutics that has licensed cadherin 11 small molecule inhibitor patents from Georgetown University. A.M., B.A., and A.A.Q. are employees of, and receive remuneration from Fluidigm Corporation. Other authors declare no potential conflict of interest.
Figures


References
-
- Rahib L, Smith BD, Aizenberg R, et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 2014;74:2913–2921. - PubMed
-
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011;364:1817–1825. - PubMed
-
- American Cancer Society. Cancer Facts & Figures 2020. Atlanta: American Cancer Society; 2020:1–76.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

