A prefrontal-bed nucleus of the stria terminalis circuit limits fear to uncertain threat
- PMID: 33319747
- PMCID: PMC7899651
- DOI: 10.7554/eLife.60812
A prefrontal-bed nucleus of the stria terminalis circuit limits fear to uncertain threat
Abstract
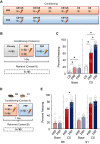
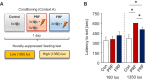
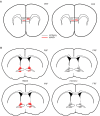
In many cases of trauma, the same environmental stimuli that become associated with aversive events are experienced on other occasions without adverse consequence. We examined neural circuits underlying partially reinforced fear (PRF), whereby mice received tone-shock pairings on half of conditioning trials. Tone-elicited freezing was lower after PRF conditioning than fully reinforced fear (FRF) conditioning, despite an equivalent number of tone-shock pairings. PRF preferentially activated medial prefrontal cortex (mPFC) and bed nucleus of the stria terminalis (BNST). Chemogenetic inhibition of BNST-projecting mPFC neurons increased PRF, not FRF, freezing. Multiplexing chemogenetics with in vivo neuronal recordings showed elevated infralimbic cortex (IL) neuronal activity during CS onset and freezing cessation; these neural correlates were abolished by chemogenetic mPFC→BNST inhibition. These data suggest that mPFC→BNST neurons limit fear to threats with a history of partial association with an aversive stimulus, with potential implications for understanding the neural basis of trauma-related disorders.
Keywords: amygdala; anxiety; fear; mouse; neuroscience; ptsd; stress; trauma.
Plain language summary
While walking home alone late one night, you hear footsteps behind you. Your heart starts to beat faster as you wonder whether someone might be following you. Being able to identify and evade threats is essential for survival. A key part of this process is learning to recognize signals that predict potential danger: the sound of footsteps behind you, for example. But many such cues are unreliable. The person behind you might simply be heading in the same general direction as you. And if you spend too much time and energy responding to such false alarms, you may struggle to complete other essential tasks. To be useful, responses to cues that signal potential threats must thus be proportionate to the likelihood that danger is actually present. By studying threat detection in mice, Glover et al. have identified a brain circuit that helps ensure that this is the case. Two groups of mice learned to fear a tone that predicted the delivery of a mild footshock. In one group of animals, the tone was followed by a shock on every trial (it was said to be ‘fully reinforced’). But in the other group, the tone was followed by a shock on only 50% of trials (‘partially reinforced’). After training, both groups of mice froze whenever they heard the tone – freezing being a typical fear response in rodents. But the animals trained with the partially reinforced tone showed less freezing than their counterparts in the fully reinforced group. Moreover, freezing in response to the partially reinforced tone was accompanied by activity in a specific neural pathway connecting the frontal part of the brain to an area called the bed nucleus of the stria terminalis. Inhibiting this pathway made mice respond to the partially reinforced tone as though it had been reinforced on every trial. This suggests that activity in this pathway helps dampen responses to unpredictable threat cues. In people with anxiety disorders, cues that become associated with unpleasant events can trigger anxiety symptoms, even if the association is unreliable. The findings of Glover et al. suggest that reduced activity of circuits that constrain excessive responses to threats might contribute to anxiety disorders.
Conflict of interest statement
LG, KM, MB, SS, NR, SO, TY, AP, MN, LH, AH No competing interests declared
Figures








References
-
- Asok A, Draper A, Hoffman AF, Schulkin J, Lupica CR, Rosen JB. Optogenetic silencing of a corticotropin-releasing factor pathway from the central amygdala to the bed nucleus of the stria terminalis disrupts sustained fear. Molecular Psychiatry. 2018;23:914–922. doi: 10.1038/mp.2017.79. - DOI - PMC - PubMed
-
- Breitfeld T, Bruning JE, Inagaki H, Takeuchi Y, Kiyokawa Y, Fendt M. Temporary inactivation of the anterior part of the bed nucleus of the stria terminalis blocks alarm pheromone-induced defensive behavior in rats. Frontiers in Neuroscience. 2015;9:321. doi: 10.3389/fnins.2015.00321. - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

