Anionic Exchange Membrane for Photo-Electrolysis Application
- PMID: 33333931
- PMCID: PMC7765393
- DOI: 10.3390/polym12122991
Anionic Exchange Membrane for Photo-Electrolysis Application
Abstract
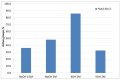
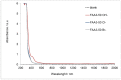
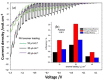
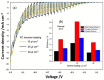
Tandem photo-electro-chemical cells composed of an assembly of a solid electrolyte membrane and two low-cost photoelectrodes have been developed to generate green solar fuel from water-splitting. In this regard, an anion-exchange polymer-electrolyte membrane, able to separate H2 evolved at the photocathode from O2 at the photoanode, was investigated in terms of ionic conductivity, corrosion mitigation, and light transmission for a tandem photo-electro-chemical configuration. The designed anionic membranes, based on polysulfone polymer, contained positive fixed functionalities on the side chains of the polymeric network, particularly quaternary ammonium species counterbalanced by hydroxide anions. The membrane was first investigated in alkaline solution, KOH or NaOH at different concentrations, to optimize the ion-exchange process. Exchange in 1M KOH solution provided high conversion of the groups, a high ion-exchange capacity (IEC) value of 1.59 meq/g and a hydroxide conductivity of 25 mS/cm at 60 °C for anionic membrane. Another important characteristic, verified for hydroxide membrane, was its transparency above 600 nm, thus making it a good candidate for tandem cell applications in which the illuminated photoanode absorbs the highest-energy photons (< 600 nm), and photocathode absorbs the lowest-energy photons. Furthermore, hydrogen crossover tests showed a permeation of H2 through the membrane of less than 0.1%. Finally, low-cost tandem photo-electro-chemical cells, formed by titanium-doped hematite and ionomer at the photoanode and cupric oxide and ionomer at the photocathode, separated by a solid membrane in OH form, were assembled to optimize the influence of ionomer-loading dispersion. Maximum enthalpy (1.7%), throughput (2.9%), and Gibbs energy efficiencies (1.3%) were reached by using n-propanol/ethanol (1:1 wt.) as solvent for ionomer dispersion and with a 25 µL cm-2 ionomer loading for both the photoanode and the photocathode.
Keywords: anion-exchange membrane; ionic conductivity; ionomer; photo-electro-chemical applications; tandem cell.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Wu Y., Li C., Tian Z., Sun J. Solar-driven integrated energy systems: State of the art and challenges. J. Power Sources. 2020;478:228762. doi: 10.1016/j.jpowsour.2020.228762. - DOI
-
- Yang W., Tavner P.J., Crabtree C.J., Feng Y., Qiu Y. Wind turbine condition monitoring: Technical and commercial challenges. Wind Energ. 2014;17:673–693. doi: 10.1002/we.1508. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

