Dissecting the role of CB1 and CB2 receptors in cannabinoid reward versus aversion using transgenic CB1- and CB2-knockout mice
- PMID: 33334652
- PMCID: PMC7854511
- DOI: 10.1016/j.euroneuro.2020.11.019
Dissecting the role of CB1 and CB2 receptors in cannabinoid reward versus aversion using transgenic CB1- and CB2-knockout mice
Abstract
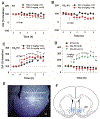
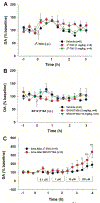
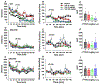
Cannabinoids produce both rewarding and aversive effects in humans and experimental animals. However, the mechanisms underlying these conflicting findings are unclear. Here we examined the potential involvement of CB1 and CB2 receptors in cannabinoid action using transgenic CB1-knockout (CB1-KO) and CB2-knockout (CB2-KO) mice. We found that Δ9-tetrahydrocannabinol (Δ9-THC) induced conditioned place preference at a low dose (1 mg/kg) in WT mice that was attenuated by deletion of the CB1 receptor. At 5 mg/kg, no subjective effects of Δ9-THC were detected in WT mice, but CB1-KO mice exhibited a trend towards place aversion and CB2-KO mice developed significant place preferences. This data suggests that activation of the CB1 receptor is rewarding, while CB2R activation is aversive. We then examined the nucleus accumbens (NAc) dopamine (DA) response to Δ9-THC using in vivo microdialysis. Unexpectedly, Δ9-THC produced a dose-dependent decrease in extracellular DA in WT mice, that was potentiated in CB1-KO mice. However, in CB2-KO mice Δ9-THC produced a dose-dependent increase in extracellular DA, suggesting that activation of the CB2R inhibits DA release in the NAc. In contrast, Δ9-THC, when administered systemically or locally into the NAc, failed to alter extracellular DA in rats. Lastly, we examined the locomotor response to Δ9-THC. Both CB1 and CB2 receptor mechanisms were shown to underlie Δ9-THC-induced hypolocomotion. These findings indicate that Δ9-THC's variable subjective effects reflect differential activation of cannabinoid receptors. Specifically, the opposing actions of CB1 and CB2 receptors regulate cannabis reward and aversion, with CB2-mediated effects predominant in mice.
Keywords: Aversion; CB(1) receptor; CB(2) receptor; Dopamine; Reward; Δ(9)-Tetrahydrocannabinol (Δ(9)-THC).
Published by Elsevier B.V.
Conflict of interest statement
Conflict of Interest The authors declare that there is no any personal financial conflict of interest.
Figures
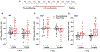

Similar articles
-
Cannabinoid CB1 Receptors Are Expressed in a Subset of Dopamine Neurons and Underlie Cannabinoid-Induced Aversion, Hypoactivity, and Anxiolytic Effects in Mice.J Neurosci. 2023 Jan 18;43(3):373-385. doi: 10.1523/JNEUROSCI.1493-22.2022. Epub 2022 Dec 14. J Neurosci. 2023. PMID: 36517243 Free PMC article.
-
Different receptor mechanisms underlying phytocannabinoid- versus synthetic cannabinoid-induced tetrad effects: Opposite roles of CB1 /CB2 versus GPR55 receptors.Br J Pharmacol. 2020 Apr;177(8):1865-1880. doi: 10.1111/bph.14958. Epub 2020 Feb 11. Br J Pharmacol. 2020. PMID: 31877572 Free PMC article.
-
Cannabinoid CB1 and CB2 receptor mechanisms underlie cannabis reward and aversion in rats.Br J Pharmacol. 2019 May;176(9):1268-1281. doi: 10.1111/bph.14625. Epub 2019 Apr 3. Br J Pharmacol. 2019. PMID: 30767215 Free PMC article.
-
The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin.Br J Pharmacol. 2008 Jan;153(2):199-215. doi: 10.1038/sj.bjp.0707442. Epub 2007 Sep 10. Br J Pharmacol. 2008. PMID: 17828291 Free PMC article. Review.
-
Distinct pharmacology and metabolism of K2 synthetic cannabinoids compared to Δ(9)-THC: mechanism underlying greater toxicity?Life Sci. 2014 Feb 27;97(1):45-54. doi: 10.1016/j.lfs.2013.09.017. Epub 2013 Sep 29. Life Sci. 2014. PMID: 24084047 Free PMC article. Review.
Cited by
-
Discordant Effects of Cannabinoid 2 Receptor Antagonism/Inverse Agonism During Adolescence on Pavlovian and Instrumental Reward Learning in Adult Male Rats.Front Synaptic Neurosci. 2021 Aug 30;13:732402. doi: 10.3389/fnsyn.2021.732402. eCollection 2021. Front Synaptic Neurosci. 2021. PMID: 34526887 Free PMC article.
-
Effects of β -caryophyllene, A Dietary Cannabinoid, in Animal Models of Drug Addiction.Curr Neuropharmacol. 2023;21(2):213-218. doi: 10.2174/1570159X20666220927115811. Curr Neuropharmacol. 2023. PMID: 36173065 Free PMC article.
-
Cannabinoid CB1 Receptors Are Expressed in a Subset of Dopamine Neurons and Underlie Cannabinoid-Induced Aversion, Hypoactivity, and Anxiolytic Effects in Mice.J Neurosci. 2023 Jan 18;43(3):373-385. doi: 10.1523/JNEUROSCI.1493-22.2022. Epub 2022 Dec 14. J Neurosci. 2023. PMID: 36517243 Free PMC article.
-
Receptor mechanisms underlying the CNS effects of cannabinoids: CB1 receptor and beyond.Adv Pharmacol. 2022;93:275-333. doi: 10.1016/bs.apha.2021.10.006. Epub 2021 Dec 13. Adv Pharmacol. 2022. PMID: 35341569 Free PMC article.
-
Quantifying conditioned place preference: a review of current analyses and a proposal for a novel approach.Front Behav Neurosci. 2023 Aug 24;17:1256764. doi: 10.3389/fnbeh.2023.1256764. eCollection 2023. Front Behav Neurosci. 2023. PMID: 37693282 Free PMC article.
References
-
- Aracil-Fernandez A, Trigo JM, Garcia-Gutierrez MS, Ortega-Alvaro A, Ternianov A, Navarro D, Robledo P, Berbel P, Maldonado R, Manzanares J, 2012. Decreased cocaine motor sensitization and self-administration in mice overexpressing cannabinoid CB(2) receptors. Neuropsychopharmacology 37, 1749–1763. - PMC - PubMed
-
- Beninger RJ, 1983. The role of dopamine in locomotor activity and learning. Brain Research Reviews 6, 173–196. - PubMed
-
- Braida D, Iosue S, Pegorini S, Sala M, 2004. Delta9-tetrahydrocannabinol-induced conditioned place preference and intracerebroventricular self-administration in rats. European Journal of Pharmacology 506, 63–69. - PubMed
-
- Buckley NE, McCoy KL, Mezey E, Bonner T, Zimmer A, Felder CC, Glass M, Zimmer A, 2000. Immunomodulation by cannabinoids is absent in mice deficient for the cannabinoid CB(2) receptor. Eur J Pharmacol 396, 141–149. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

