Therapeutic Potential of Exosomes in Pulmonary Fibrosis
- PMID: 33343360
- PMCID: PMC7746877
- DOI: 10.3389/fphar.2020.590972
Therapeutic Potential of Exosomes in Pulmonary Fibrosis
Abstract
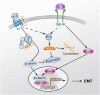
Pulmonary fibrosis is closely associated with the recruitment of fibroblasts from capillary vessels with damaged endothelial cells, the epithelial mesenchymal transition (EMT) of type II alveolar epithelial cells, and the transformation of fibroblasts to myofibroblasts. Recent studies suggest that EMT is a key factor in the pathogenesis of pulmonary fibrosis, as the disruption of EMT-related effector molecules can inhibit the occurrence and development of PF. With the numerous advancements made in molecular biology in recent years, researchers have discovered that exosomes and their cargos, such as miRNAs, lncRNAs, and proteins, can promote or inhibit the EMT, modulate the transformation of fibroblasts into myofibroblasts, contribute to the proliferation of fibroblasts and promote immunoregulatory and mitochondrial damage during pulmonary fibrosis. Exosomes are key factors regulating the differentiation of bone marrow mesenchymal stem cells (BMSCs) into myofibroblasts. Interestingly, exosomes derived from BMSCs under pathological and physiological conditions may promote or inhibit the EMT of type II alveolar epithelial cells and the transformation of fibroblasts into myofibroblasts to regulate pulmonary fibrosis. Thus, exosomes may become a new direction in the study of drugs for the treatment of pulmonary fibrosis.
Keywords: bone marrow mesenchymal stem cell; epithelial mesenchymal transition; exosome; myofibroblast; pulmonary fibrosis; type II alveolar epithelial cell.
Copyright © 2020 Xie and Zeng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Snail-mediated partial epithelial mesenchymal transition augments the differentiation of local lung myofibroblast.Chemosphere. 2021 Mar;267:128870. doi: 10.1016/j.chemosphere.2020.128870. Epub 2020 Nov 4. Chemosphere. 2021. PMID: 33172668
-
Exosomes derived from bone marrow mesenchymal stem cells reverse epithelial-mesenchymal transition potentially via attenuating Wnt/β-catenin signaling to alleviate silica-induced pulmonary fibrosis.Toxicol Mech Methods. 2021 Nov;31(9):655-666. doi: 10.1080/15376516.2021.1950250. Epub 2021 Aug 12. Toxicol Mech Methods. 2021. PMID: 34225584
-
Carbon nanotubes promote alveolar macrophages toward M2 polarization mediated epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation.Nanotoxicology. 2021 Jun;15(5):588-604. doi: 10.1080/17435390.2021.1905098. Epub 2021 Apr 10. Nanotoxicology. 2021. PMID: 33840345
-
Recent advances in molecular targets and treatment of idiopathic pulmonary fibrosis: focus on TGFbeta signaling and the myofibroblast.Curr Med Chem. 2009;16(11):1400-17. doi: 10.2174/092986709787846497. Curr Med Chem. 2009. PMID: 19355895 Review.
-
Molecular Pathogenesis of Pulmonary Fibrosis, with Focus on Pathways Related to TGF-β and the Ubiquitin-Proteasome Pathway.Int J Mol Sci. 2021 Jun 5;22(11):6107. doi: 10.3390/ijms22116107. Int J Mol Sci. 2021. PMID: 34198949 Free PMC article. Review.
Cited by
-
Exosomal Micro-RNAs as Intercellular Communicators in Idiopathic Pulmonary Fibrosis.Int J Mol Sci. 2022 Sep 20;23(19):11047. doi: 10.3390/ijms231911047. Int J Mol Sci. 2022. PMID: 36232350 Free PMC article. Review.
-
The Role of Macrophages in Lung Fibrosis and the Signaling Pathway.Cell Biochem Biophys. 2024 Jun;82(2):479-488. doi: 10.1007/s12013-024-01253-5. Epub 2024 Mar 27. Cell Biochem Biophys. 2024. PMID: 38536578 Review.
-
Phosphorylation of FOXN3 by NEK6 promotes pulmonary fibrosis through Smad signaling.Nat Commun. 2025 Feb 21;16(1):1865. doi: 10.1038/s41467-025-56922-7. Nat Commun. 2025. PMID: 39984467 Free PMC article.
-
Exosomes in pathogenesis, diagnosis, and treatment of pulmonary fibrosis.Front Pharmacol. 2022 Aug 25;13:927653. doi: 10.3389/fphar.2022.927653. eCollection 2022. Front Pharmacol. 2022. PMID: 36091791 Free PMC article. Review.
-
Exosomal miR-17-5p from human embryonic stem cells prevents pulmonary fibrosis by targeting thrombospondin-2.Stem Cell Res Ther. 2023 Sep 4;14(1):234. doi: 10.1186/s13287-023-03449-7. Stem Cell Res Ther. 2023. PMID: 37667335 Free PMC article.
References
-
- Bandeira E., Oliveira H., Silva J. D., Menna-Barreto R. F. S., Takyia C. M., Suk J. S., et al. (2018). Therapeutic effects of adipose-tissue-derived mesenchymal stromal cells and their extracellular vesicles in experimental silicosis. Respir. Res. 19 (1), 104 10.1186/s12931-018-0802-3 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

