Light-Mediated Signaling and Metabolic Changes Coordinate Stomatal Opening and Closure
- PMID: 33343603
- PMCID: PMC7746640
- DOI: 10.3389/fpls.2020.601478
Light-Mediated Signaling and Metabolic Changes Coordinate Stomatal Opening and Closure
Abstract
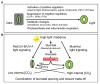
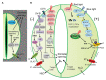
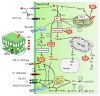
Stomata are valves on the leaf surface controlling carbon dioxide (CO2) influx for photosynthesis and water loss by transpiration. Thus, plants have to evolve elaborate mechanisms controlling stomatal aperture to allow efficient photosynthesis while avoid excessive water loss. Light is not only the energy source for photosynthesis but also an important signal regulating stomatal movement during dark-to-light transition. Our knowledge concerning blue and red light signaling and light-induced metabolite changes that contribute to stomatal opening are accumulating. This review summarizes recent advances on the signaling components that lie between the perception of blue/red light and activation of the PM H+-ATPases, and on the negative regulation of stomatal opening by red light-activated phyB signaling and ultraviolet (UV-B and UV-A) irradiation. Besides, light-regulated guard cell (GC)-specific metabolic levels, mesophyll-derived sucrose, and CO2 concentration within GCs also play dual roles in stomatal opening. Thus, light-induced stomatal opening is tightly accompanied by brake mechanisms, allowing plants to coordinate carbon gain and water loss. Knowledge on the mechanisms regulating the trade-off between stomatal opening and closure may have potential applications toward generating superior crops with improved water use efficiency (CO2 gain vs. water loss).
Keywords: Arabidopsis thaliana; guard cell metabolism; light signaling; negative mechanism; stomatal movement; trade-off.
Copyright © 2020 Yang, Li, Kong, Guo and Wei.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Azoulay-Shemer T., Palomares A., Bagheri A., Israelsson-Nordstrom M., Engineer C. B., Bargmann B. O. R., et al. (2015). Guard cell photosynthesis is critical for stomatal turgor production, yet does not directly mediate CO2‐ and ABA-induced stomatal closing. Plant J. 83, 567–581. 10.1111/tpj.12916, PMID: - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

