Neonatal Hyperoxia Downregulates Claudin-4, Occludin, and ZO-1 Expression in Rat Kidney Accompanied by Impaired Proximal Tubular Development
- PMID: 33343804
- PMCID: PMC7725566
- DOI: 10.1155/2020/2641461
Neonatal Hyperoxia Downregulates Claudin-4, Occludin, and ZO-1 Expression in Rat Kidney Accompanied by Impaired Proximal Tubular Development
Abstract
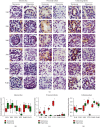
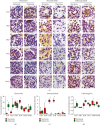
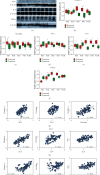
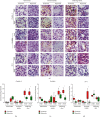
Hyperoxia is essential to manage in preterm infants but causes injury to immature kidney. Previous study indicates that hyperoxia causes oxidative damage to neonatal kidney and impairs renal development. However, the underlying mechanisms by which neonatal hyperoxia effects on immature kidney still need to be elucidated. Tight junction, among which the representative proteins are claudin-4, occludin, and ZO-1, plays a crucial role in nephrogenesis and maintaining renal function. Inflammatory cytokines are involved in the pleiotropic regulation of tight junction proteins. Here, we investigated how neonatal hyperoxia affected the expression of key tight junction proteins and inflammatory factors (IL-6 and TNF-α) in the developing rat kidneys and elucidated their correlation with renal injury. We found claudin-4, occludin, and zonula occludens-1 (ZO-1) expression in proximal tubules was significantly downregulated after neonatal hyperoxia. The expression of these tight junction proteins was positively correlated with that of IL-6 and TNF-α, while claudin-4 expression was positively correlated with injury score of proximal tubules in mature kidneys. These findings indicated that impaired expression of tight junction proteins in kidney might be a potential mechanism of hyperoxia-induced nephrogenic disorders. It provides new insights to further study oxidative renal injury and development disorders and will be helpful for seeking potential therapeutics for hyperoxia-induced renal injury in the future.
Copyright © 2020 Xuewen Xu et al.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Figures

Similar articles
-
Hyperoxia disrupts pulmonary epithelial barrier in newborn rats via the deterioration of occludin and ZO-1.Respir Res. 2012 May 4;13(1):36. doi: 10.1186/1465-9921-13-36. Respir Res. 2012. PMID: 22559818 Free PMC article.
-
Proximal Tubular Development Is Impaired with Downregulation of MAPK/ERK Signaling, HIF-1α, and Catalase by Hyperoxia Exposure in Neonatal Rats.Oxid Med Cell Longev. 2019 Aug 28;2019:9219847. doi: 10.1155/2019/9219847. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31558952 Free PMC article.
-
Renal ischemia-reperfusion injury causes intercalated cell-specific disruption of occludin in the collecting duct.Histochem Cell Biol. 2011 Dec;136(6):637-47. doi: 10.1007/s00418-011-0881-4. Epub 2011 Nov 3. Histochem Cell Biol. 2011. PMID: 22048282 Free PMC article.
-
MicroRNA Regulation of Endothelial Junction Proteins and Clinical Consequence.Mediators Inflamm. 2016;2016:5078627. doi: 10.1155/2016/5078627. Epub 2016 Nov 24. Mediators Inflamm. 2016. PMID: 27999452 Free PMC article. Review.
-
Kidney Injuries and Evolution of Chronic Kidney Diseases Due to Neonatal Hyperoxia Exposure Based on Animal Studies.Int J Mol Sci. 2022 Jul 31;23(15):8492. doi: 10.3390/ijms23158492. Int J Mol Sci. 2022. PMID: 35955627 Free PMC article. Review.
Cited by
-
Anti-Tn Monoclonal Antibody Ameliorates Hyperoxia-Induced Kidney Injury by Suppressing Oxidative Stress and Inflammation in Neonatal Mice.Mediators Inflamm. 2021 Oct 21;2021:1180543. doi: 10.1155/2021/1180543. eCollection 2021. Mediators Inflamm. 2021. PMID: 34720748 Free PMC article.
-
The danger of hyperoxia on the rat kidneys: is tadalafil a real shield?Int Urol Nephrol. 2023 Feb;55(2):241-247. doi: 10.1007/s11255-022-03416-w. Epub 2022 Nov 29. Int Urol Nephrol. 2023. PMID: 36443608 Free PMC article.
-
Hyperoxia and Acute Kidney Injury: A Tale of Oxygen and the Kidney.Semin Nephrol. 2022 May;42(3):151282. doi: 10.1016/j.semnephrol.2022.10.008. Epub 2022 Nov 18. Semin Nephrol. 2022. PMID: 36404211 Free PMC article. Review.
-
The effect of NF-kB and MAPK mediated Proinflammatory microenvironment on renal aging and amyloid deposition in elder rats.Sci Rep. 2025 Aug 18;15(1):30188. doi: 10.1038/s41598-025-14559-y. Sci Rep. 2025. PMID: 40825962 Free PMC article.
-
Matrix Metalloproteinase-10 in Kidney Injury Repair and Disease.Int J Mol Sci. 2022 Feb 15;23(4):2131. doi: 10.3390/ijms23042131. Int J Mol Sci. 2022. PMID: 35216251 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

