Fusogenic oncolytic vaccinia virus enhances systemic antitumor immune response by modulating the tumor microenvironment
- PMID: 33348052
- PMCID: PMC8116573
- DOI: 10.1016/j.ymthe.2020.12.024
Fusogenic oncolytic vaccinia virus enhances systemic antitumor immune response by modulating the tumor microenvironment
Abstract
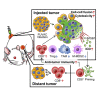
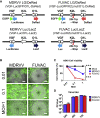
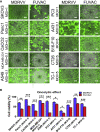
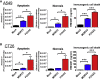
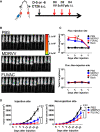
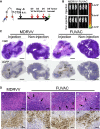
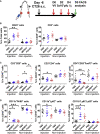
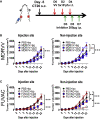
Oncolytic viruses induce antitumor immunity following direct viral oncolysis. However, their therapeutic effects are limited in distant untreated tumors because their antitumor function depends on indirect antitumor immunity. Here, we generated a novel fusogenic oncolytic vaccinia virus (FUVAC) and compared its antitumor activity with that of its parental non-fusogenic virus. Compared with the parent, FUVAC exerted the cytopathic effect and induced immunogenic cell death in human and murine cancer cells more efficiently. In a bilateral tumor-bearing syngeneic mouse model, FUVAC administration significantly inhibited tumor growth in both treated and untreated tumors. However, its antitumor effects were completely suppressed by CD8+ T cell depletion. Notably, FUVAC reduced the number of tumor-associated immune-suppressive cells in treated tumors, but not in untreated tumors. Mice treated with FUVAC before an immune checkpoint inhibitor (ICI) treatment achieved complete response (CR) in both treated and untreated tumors, whereas ICI alone did not show antitumor activity. Mice achieving CR rejected rechallenge with the same tumor cells, suggesting establishment of a long-term tumor-specific immune memory. Thus, FUVAC improves the tumor immune microenvironment and enhances systemic antitumor immunity, suggesting that, alone and in combination with ICI, it is a novel immune modulator for overcoming oncolytic virus-resistant tumors.
Keywords: antitumor immunity; cell membrane fusion; immune checkpoint blockade; oncolytic function; vaccinia virus.
Copyright © 2020 The American Society of Gene and Cell Therapy. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Histone deacetylase inhibitor boosts anticancer potential of fusogenic oncolytic vaccinia virus by enhancing cell-cell fusion.Cancer Sci. 2024 Feb;115(2):600-610. doi: 10.1111/cas.16032. Epub 2023 Nov 30. Cancer Sci. 2024. PMID: 38037288 Free PMC article.
-
A systemically deliverable Vaccinia virus with increased capacity for intertumoral and intratumoral spread effectively treats pancreatic cancer.J Immunother Cancer. 2021 Jan;9(1):e001624. doi: 10.1136/jitc-2020-001624. J Immunother Cancer. 2021. PMID: 33500259 Free PMC article.
-
Intratumoral expression of interleukin 23 variants using oncolytic vaccinia virus elicit potent antitumor effects on multiple tumor models via tumor microenvironment modulation.Theranostics. 2021 May 3;11(14):6668-6681. doi: 10.7150/thno.56494. eCollection 2021. Theranostics. 2021. PMID: 34093846 Free PMC article.
-
Using oncolytic viruses to ignite the tumour immune microenvironment in bladder cancer.Nat Rev Urol. 2021 Sep;18(9):543-555. doi: 10.1038/s41585-021-00483-z. Epub 2021 Jun 28. Nat Rev Urol. 2021. PMID: 34183833 Review.
-
Oncolytic viruses for cancer immunotherapy.J Hematol Oncol. 2020 Jun 29;13(1):84. doi: 10.1186/s13045-020-00922-1. J Hematol Oncol. 2020. PMID: 32600470 Free PMC article. Review.
Cited by
-
Mesenchymal Stem Cells Carrying Viral Fusogenic Protein p14 to Treat Solid Tumors by Inducing Cell-Cell Fusion and Immune Activation.Research (Wash D C). 2025 Jan 27;8:0594. doi: 10.34133/research.0594. eCollection 2025. Research (Wash D C). 2025. PMID: 39872128 Free PMC article.
-
Oncolytic Vaccinia Virus in Lung Cancer Vaccines.Vaccines (Basel). 2022 Feb 4;10(2):240. doi: 10.3390/vaccines10020240. Vaccines (Basel). 2022. PMID: 35214699 Free PMC article. Review.
-
A new MVA ancestor-derived oncolytic vaccinia virus induces immunogenic tumor cell death and robust antitumor immune responses.Mol Ther. 2024 Jul 3;32(7):2406-2422. doi: 10.1016/j.ymthe.2024.05.014. Epub 2024 May 11. Mol Ther. 2024. PMID: 38734899 Free PMC article.
-
Histone deacetylase inhibitor boosts anticancer potential of fusogenic oncolytic vaccinia virus by enhancing cell-cell fusion.Cancer Sci. 2024 Feb;115(2):600-610. doi: 10.1111/cas.16032. Epub 2023 Nov 30. Cancer Sci. 2024. PMID: 38037288 Free PMC article.
-
Oncolytic Viruses as a Novel Therapeutic Approach for Colorectal Cancer: Mechanisms, Current Advances, and Future Directions.Cancers (Basel). 2025 May 31;17(11):1854. doi: 10.3390/cancers17111854. Cancers (Basel). 2025. PMID: 40507337 Free PMC article. Review.
References
-
- Andtbacka R.H., Ross M., Puzanov I., Milhem M., Collichio F., Delman K.A., Amatruda T., Zager J.S., Cranmer L., Hsueh E. Patterns of clinical response with talimogene laherparepvec (T-VEC) in patients with melanoma treated in the OPTiM Phase III clinical trial. Ann. Surg. Oncol. 2016;23:4169–4177. - PMC - PubMed
-
- Kaufman H.L., Amatruda T., Reid T., Gonzalez R., Glaspy J., Whitman E., Harrington K., Nemunaitis J., Zloza A., Wolf M., Senzer N.N. Systemic versus local responses in melanoma patients treated with talimogene laherparepvec from a multi-institutional phase II study. J. Immunother. Cancer. 2016;4:12. - PMC - PubMed
-
- Park B.H., Hwang T., Liu T.C., Sze D.Y., Kim J.S., Kwon H.C., Oh S.Y., Han S.Y., Yoon J.H., Hong S.H. Use of a targeted oncolytic poxvirus, JX-594, in patients with refractory primary or metastatic liver cancer: a phase I trial. Lancet Oncol. 2008;9:533–542. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

