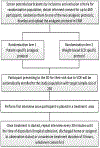
Trial design of comparing patient-specific versus weight-based protocols to treat vaso-occlusive episodes in sickle cell disease (COMPARE-VOE)
- PMID: 33348066
- PMCID: PMC7954959
- DOI: 10.1016/j.cct.2020.106252
Trial design of comparing patient-specific versus weight-based protocols to treat vaso-occlusive episodes in sickle cell disease (COMPARE-VOE)
Abstract
Objectives: Painful vaso-occlusive episodes (VOE) are the most common reason for emergency department (ED) visits experienced by patients with sickle cell disease (SCD). The National Heart, Lung and Blood Institute (NHLBI) evidence-based recommendations for VOE treatment are based primarily on expert opinion. In this randomized controlled trial (RCT), we will compare changes in pain scores between patients randomized to a patient-specific analgesic protocol versus those randomized to a weight-based analgesic protocol, as recommended by the NHLBI guidelines.
Methods: We report the rationale and design of a multi-site, phase III, single-blinded, RCT to be conducted in six EDs in the United States. Eligible participants will be randomized after providing consent, anticipating 50% of those randomized would have an ED visit during the enrollment period. A total of 230 participants with one VOE ED visit provides sufficient power to detect a clinically significant difference in pain score reductions of 14 between groups with 0.05 type I error. Uniquely, this trial randomizes participants in a larger population than the study population, given the impossibility of consenting and randomizing participants during emergencies. The primary endpoint is the change in pain scores in the ED from time of placement in treatment area to time of disposition (hospitalization, discharged home, or assigned to observation status) or a maximum treatment duration of 6 hours. Additional outcomes include hospitalizations and ED visits seven days post enrollment, side effects, and safety assessments.
Conclusions: The COMPARE-VOE study design will provide high-level evidence to support the NHLBI VOE treatment guidelines.
Keywords: Clinical trial; Emergency department; Pain management protocol; Sickle cell disease; Vaso-occlusive crisis; Vaso-occlusive episode.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
CONFLICT OF INTEREST STATEMENT
Stephanie Ibemere has received postdoctoral fellowship salary support from the NHLBI.
Sarah Dubbs declares no conflict of interest.
Huiman Barnhart declares no conflict of interest
Jacqueline Brown declares no conflict of interest.
Caroline Freiermuth has received a quality improvement grant from Pfizer, administered by the American College of Emergency Physicians. In addition, she was a site investigator and received research funds from Pfizer for the Phase 3 trial on safety and efficacy on Rivipansal. All research funds were distributed to the University of Cincinnati and not directly to the investigator.
Patricia Kavanagh declares no conflicts of interest.
Judith Paice declares no conflicts of interest.
John Strouse has received research support through his institution from Takedea Pharmaceutical Company Limited.
R. Gentry Wilkerson has received a grant from Pfizer and the American College of Emergency Physicians and has received research support from Pfizer and Prolong Pharmaceuticals.
Paula Tanabe declares no conflict of interest.
References
-
- Jiang HJ, Barrett ML, Sheng M. Characteristics of Hospital Stays for Nonelderly Medicaid Super-Utilizers, 2012: Statistical Brief #184. In: Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Agency for Healthcare Research and Quality (US); 2006. Accessed April 26, 2020. http://www.ncbi.nlm.nih.gov/books/NBK269028/ - PubMed
-
- Benjamin L, Dampier C, Jacox A, Odesina V, Phoenix D, Shapiro B. Guideline for the Mangement of Acute and Chronic Pain in Sickle-Cell Disease. Glenview IL Am Pain Soc. 1999;(1).
-
- National Heart, Lung, and Blood Institute. Evidence-Based Management of Sickle Cell Disease: Expert Panel, 2014. Dep Health Hum Serv Natl Inst Health Natl Heart Lung Blood Inst. Published online 2014:161.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical