Gangliosides in Podocyte Biology and Disease
- PMID: 33348903
- PMCID: PMC7766259
- DOI: 10.3390/ijms21249645
Gangliosides in Podocyte Biology and Disease
Abstract
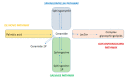
Gangliosides constitute a subgroup of glycosphingolipids characterized by the presence of sialic acid residues in their structure. As constituents of cellular membranes, in particular of raft microdomains, they exert multiple functions, some of them capital in cell homeostasis. Their presence in cells is tightly regulated by a balanced expression and function of the enzymes responsible for their biosynthesis, ganglioside synthases, and their degradation, glycosidases. The dysregulation of their abundance results in rare and common diseases. In this review, we make a point on the relevance of gangliosides and some of their metabolic precursors, such as ceramides, in the function of podocytes, the main cellular component of the glomerular filtration barrier, as well as their implications in podocytopathies. The results presented in this review suggest the pertinence of clinical lipidomic studies targeting these metabolites.
Keywords: ceramide; glomerulopathies; glomerulus; glycosphingolipids; kidney; nephrotic syndrome; podocytopathies; rafts.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Mauri L., Sonnino S., Prinetti A. Gangliosides. Volume 1804. Humana Press; New York, NY, USA: 2018. Chemical and physicochemical properties of gangliosides; pp. 1–17. - PubMed
-
- Inokuchi J., Inamori K.-I., Kabayama K., Nagafuku M., Uemura S., Go S., Suzuki A., Ohno I., Kanoh H., Shishido F. Biology of GM3 ganglioside. Prog. Mol. Biol. Transl. Sci. 2018;156:151–195. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

