Characterization of "Free Base" and Metal Complex Thioalkyl Porphyrazines by Magnetic Circular Dichroism and TDDFT Calculations
- PMID: 33351631
- PMCID: PMC8016196
- DOI: 10.1021/acs.jpcb.0c09277
Characterization of "Free Base" and Metal Complex Thioalkyl Porphyrazines by Magnetic Circular Dichroism and TDDFT Calculations
Abstract
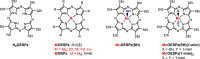
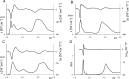
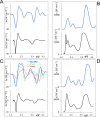
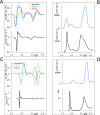
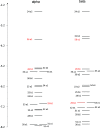
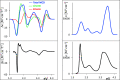
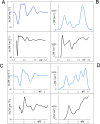
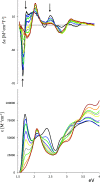
UV-vis absorption and magnetic circular dichroism (MCD) spectra of octakis thioethyl "free base" porphyrazine H2OESPz and its metal complexes MOESPz (M = Mg, Zn, Ni, Pd, Cu), as well as of [MnOESPz(SH)] were recorded. In the last case, MCD proved to have quite good sensitivity to the coordination of this complex with 1-methylimidazole (1-mim) in benzene. Time-dependent density functional theory (TDDFT) calculations were carried out for the considered porphyrazine complexes and showed good performance on comparing with MCD and UV-vis experimental spectra, even in the open-shell Cu and Mn cases. Calculations accounted for the red shift observed in the thioalkyl compounds and allowed us to reveal the role of sulfur atoms in spectroscopically relevant molecular orbitals and to highlight the importance of the conformations of the thioethyl external groups. Calculated MCD spectra of [MnOESPz(SH)] confirm the Mn(III) → Mn(II) redox process, which leads to the [Mn(OESPz)(1-mim)2] species, and the relevance of the spin state for MCD is revealed.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Characterization of tetrakis(thiadiazole)porphyrazine metal complexes by magnetic circular dichroism and magnetic circularly polarized luminescence.Chirality. 2020 Jun;32(6):808-816. doi: 10.1002/chir.23221. Epub 2020 Mar 18. Chirality. 2020. PMID: 32189418
-
Influence of molecular geometry, exchange-correlation functional, and solvent effects in the modeling of vertical excitation energies in phthalocyanines using time-dependent density functional theory (TDDFT) and polarized continuum model TDDFT methods: can modern computational chemistry methods explain experimental controversies?J Phys Chem A. 2007 Dec 20;111(50):12901-13. doi: 10.1021/jp0759731. Epub 2007 Nov 16. J Phys Chem A. 2007. PMID: 18004829
-
Magnetic Circular Dichroism of Transition-Metal Complexes of Perfluorophenyl-N-Confused Porphyrins: Inverting Electronic Structure through a Proton.J Phys Chem A. 2017 May 18;121(19):3689-3698. doi: 10.1021/acs.jpca.7b02908. Epub 2017 May 5. J Phys Chem A. 2017. PMID: 28475333
-
Application of magnetically perturbed time-dependent density functional theory to magnetic circular dichroism. III. Temperature-dependent magnetic circular dichroism induced by spin-orbit coupling.J Chem Phys. 2008 Sep 14;129(10):104105. doi: 10.1063/1.2976568. J Chem Phys. 2008. PMID: 19044906
-
Electronic structure and spectroscopic properties of mononuclear manganese(III) Schiff base complexes: a systematic study on [Mn(acen)X] complexes by EPR, UV/vis, and MCD spectroscopy (X = Hal, NCS).Inorg Chem. 2013 Mar 4;52(5):2372-87. doi: 10.1021/ic301889e. Epub 2013 Feb 14. Inorg Chem. 2013. PMID: 23410227
Cited by
-
MCD and MCPL Characterization of Luminescent Si(IV) and P(V) Tritolylcorroles: The Role of Coordination Number.ACS Omega. 2021 Sep 29;6(40):26659-26671. doi: 10.1021/acsomega.1c04028. eCollection 2021 Oct 12. ACS Omega. 2021. PMID: 34661019 Free PMC article.
References
-
- Walter M. G.; Rudine A. B.; Wamser C. C. Porphyrins and Phthalocyanines in Solar Photovoltaic Cells. J. Porphyrins Phthalocyanines 2010, 14, 759–792. 10.1142/S1088424610002689. - DOI
-
- Travkin V.; Yunin P.; Stuzhin P.; Pakhomov G. Characterization of Vacuum-Deposited Films of Hexachloro-Substituted Subphthalocyanines for Photovoltaic Applications. Mater. Today: Proc. 2020, 20, 12–15. 10.1016/j.matpr.2019.07.708. - DOI
-
- Drobizhev M.; Makarov N. S.; Rebane A.; de la Torre G.; Torres T. Strong Two-Photon Absorption in Push–Pull Phthalocyanines: Role of Resonance Enhancement and Permanent Dipole Moment Change upon Excitation. J. Phys. Chem. C 2008, 112, 848–859. 10.1021/jp073318n. - DOI
-
- Senge M. O.; Fazekas M.; Notaras E. G. A.; Blau W. J.; Zawadzka M.; Locos O. B.; Ni Mhuircheartaigh E. M. Nonlinear Optical Properties of Porphyrins. Adv. Mater. 2007, 19, 2737–2774. 10.1002/adma.200601850. - DOI
Publication types
LinkOut - more resources
Full Text Sources

