Effects of Propranolol on Growth, Lipids and Energy Metabolism and Oxidative Stress Response of Phaeodactylum tricornutum
- PMID: 33353054
- PMCID: PMC7766914
- DOI: 10.3390/biology9120478
Effects of Propranolol on Growth, Lipids and Energy Metabolism and Oxidative Stress Response of Phaeodactylum tricornutum
Abstract
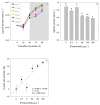
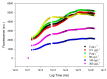
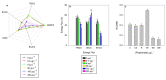
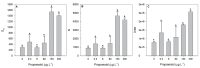
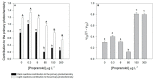
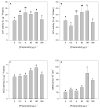
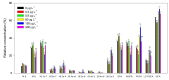
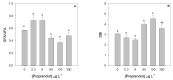
Present demographic trends suggest a rise in the contributions of human pharmaceuticals into coastal ecosystems, underpinning an increasing demand to evaluate the ecotoxicological effects and implications of drug residues in marine risk assessments. Propranolol, a non-selective β-adrenoceptor blocker, is used worldwide to treat high blood pressure conditions and other related cardiovascular conditions. Although diatoms lack β-adrenoceptors, this microalgal group presents receptor-like kinases and proteins with a functional analogy to the animal receptors and that can be targeted by propranolol. In the present work, the authors evaluated the effect of this non-selective β-adrenoceptor blocker in diatom cells using P. tricornutum as a model organism, to evaluate the potential effect of this compound in cell physiology (growth, lipids and energy metabolism and oxidative stress) and its potential relevance for marine ecosystems. Propranolol exposure leads to a significant reduction in diatom cell growth, more evident in the highest concentrations tested. This is likely due to the observed impairment of the main primary photochemistry processes and the enhancement of the mitochondrial respiratory activity. More specifically, propranolol decreased the energy transduction from photosystem II (PSII) to the electron transport chain, leading to an increase in oxidative stress levels. Cells exposed to propranolol also exhibited high-dissipated energy flux, indicating that this excessive energy is efficiently diverted, to some extent, from the photosystems, acting to prevent irreversible photoinhibition. As energy production is impaired at the PSII donor side, preventing energy production through the electron transport chain, diatoms appear to be consuming storage lipids as an energy backup system, to maintain essential cellular functions. This consumption will be attained by an increase in respiratory activity. Considering the primary oxygen production and consumption pathways, propranolol showed a significant reduction of the autotrophic O2 production and an increase in the heterotrophic mitochondrial respiration. Both mechanisms can have negative effects on marine trophic webs, due to a decrease in the energetic input from marine primary producers and a simultaneous oxygen production decrease for heterotrophic species. In ecotoxicological terms, bio-optical and fatty acid data appear as highly efficient tools for ecotoxicity assessment, with an overall high degree of classification when these traits are used to build a toxicological profile, instead of individually assessed.
Keywords: energy metabolism; pharmatoxicology; photobiology; primary producers; toxicophenomics.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


Similar articles
-
Fluoxetine Arrests Growth of the Model Diatom Phaeodactylum tricornutum by Increasing Oxidative Stress and Altering Energetic and Lipid Metabolism.Front Microbiol. 2020 Jul 31;11:1803. doi: 10.3389/fmicb.2020.01803. eCollection 2020. Front Microbiol. 2020. PMID: 32849412 Free PMC article.
-
Ecotoxicity of the lipid-lowering drug bezafibrate on the bioenergetics and lipid metabolism of the diatom Phaeodactylum tricornutum.Sci Total Environ. 2019 Feb 10;650(Pt 2):2085-2094. doi: 10.1016/j.scitotenv.2018.09.354. Epub 2018 Oct 1. Sci Total Environ. 2019. PMID: 30290350
-
Enantioselective toxicity of propranolol on marine diatoms: Assessing growth, energy metabolism and oxidative damage in Phaeodactylum tricornutum.Mar Pollut Bull. 2025 May;214:117751. doi: 10.1016/j.marpolbul.2025.117751. Epub 2025 Mar 4. Mar Pollut Bull. 2025. PMID: 40043661
-
Modelling metabolism of the diatom Phaeodactylum tricornutum.Biochem Soc Trans. 2015 Dec;43(6):1182-6. doi: 10.1042/BST20150152. Biochem Soc Trans. 2015. PMID: 26614658 Review.
-
Current trends to comprehend lipid metabolism in diatoms.Prog Lipid Res. 2018 Apr;70:1-16. doi: 10.1016/j.plipres.2018.03.001. Epub 2018 Mar 7. Prog Lipid Res. 2018. PMID: 29524459 Review.
Cited by
-
Ecotoxicological Effects of the Anionic Surfactant Sodium Dodecyl Sulfate (SDS) in Two Marine Primary Producers: Phaeodactylum tricornutum and Ulva lactuca.Toxics. 2022 Dec 13;10(12):780. doi: 10.3390/toxics10120780. Toxics. 2022. PMID: 36548613 Free PMC article.
-
Biomorphometric and hematobiochemical alterations in the juvenile african catfish Clarias gariepinus exposed to propranolol.BMC Zool. 2024 Jun 20;9(1):11. doi: 10.1186/s40850-024-00196-x. BMC Zool. 2024. PMID: 38902830 Free PMC article.
-
Artificial Intelligence Meets Marine Ecotoxicology: Applying Deep Learning to Bio-Optical Data from Marine Diatoms Exposed to Legacy and Emerging Contaminants.Biology (Basel). 2021 Sep 18;10(9):932. doi: 10.3390/biology10090932. Biology (Basel). 2021. PMID: 34571809 Free PMC article.
-
Untargeted Metabolomics Reveals Antidepressant Effects in a Marine Photosynthetic Organism: The Diatom Phaeodactylum tricornutum as a Case Study.Biology (Basel). 2022 Dec 6;11(12):1770. doi: 10.3390/biology11121770. Biology (Basel). 2022. PMID: 36552278 Free PMC article.
-
Effects of Glyphosate-Based Herbicide on Primary Production and Physiological Fitness of the Macroalgae Ulva lactuca.Toxics. 2022 Jul 28;10(8):430. doi: 10.3390/toxics10080430. Toxics. 2022. PMID: 36006109 Free PMC article.
References
-
- Martínez M.L., Intralawan A., Vázquez G., Pérez-Maqueo O., Sutton P., Landgrave R. The coasts of our world: Ecological, economic and social importance. Ecol. Econ. 2007;63:254–272. doi: 10.1016/j.ecolecon.2006.10.022. - DOI
-
- Li H. Management of coastal mega-cities—A new challenge in the 21st century. Mar. Policy. 2003;27:333–337. doi: 10.1016/S0308-597X(03)00045-9. - DOI
-
- Von Glasow R., Jickells T.D., Baklanov A., Carmichael G.R., Church T.M., Gallardo L., Hughes C., Kanakidou M., Liss P.S., Mee L., et al. Megacities and Large Urban Agglomerations in the Coastal Zone: Interactions between Atmosphere, Land, and Marine Ecosystems. Ambio. 2013;42:13–28. doi: 10.1007/s13280-012-0343-9. - DOI - PMC - PubMed
-
- Reis-Santos P., Pais M., Duarte B., Caçador I., Freitas A., Vila Pouca A.S., Barbosa J., Leston S., Rosa J., Ramos F., et al. Screening of human and veterinary pharmaceuticals in estuarine waters: A baseline assessment for the Tejo estuary. Mar. Pollut. Bull. 2018;135:1079–1084. doi: 10.1016/j.marpolbul.2018.08.036. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

