The symbiotic 'all-rounders': Partnerships between marine animals and chemosynthetic nitrogen-fixing bacteria
- PMID: 33355107
- PMCID: PMC8090883
- DOI: 10.1128/AEM.02129-20
The symbiotic 'all-rounders': Partnerships between marine animals and chemosynthetic nitrogen-fixing bacteria
Abstract
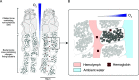
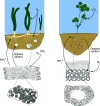
Nitrogen fixation is a widespread metabolic trait in certain types of microorganisms called diazotrophs. Bioavailable nitrogen is limited in various habitats on land and in the sea, and accordingly, a range of plant, animal, and single-celled eukaryotes have evolved symbioses with diverse diazotrophic bacteria, with enormous economic and ecological benefits. Until recently, all known nitrogen-fixing symbionts were heterotrophs such as nodulating rhizobia, or photoautotrophs such as cyanobacteria. In 2016, the first chemoautotrophic nitrogen-fixing symbionts were discovered in a common family of marine clams, the Lucinidae. Chemosynthetic nitrogen-fixing symbionts use the chemical energy stored in reduced sulfur compounds to power carbon and nitrogen fixation, making them metabolic 'all-rounders' with multiple functions in the symbiosis. This distinguishes them from heterotrophic symbionts that require a source of carbon from their host, and their chemosynthetic metabolism distinguishes them from photoautotrophic symbionts that produce oxygen, a potent inhibitor of nitrogenase. In this review, we consider evolutionary aspects of this discovery, by comparing strategies that have evolved for hosting intracellular nitrogen-fixing symbionts in plants and animals. The symbiosis between lucinid clams and chemosynthetic nitrogen-fixing bacteria also has important ecological impacts, as they form a nested symbiosis with endangered marine seagrasses. Notably, nitrogen fixation by lucinid symbionts may help support seagrass health by providing a source of nitrogen in seagrass habitats. These discoveries were enabled by new techniques for understanding the activity of microbial populations in natural environments. However, an animal (or plant) host represents a diverse landscape of microbial niches due to its structural, chemical, immune and behavioural properties. In future, methods that resolve microbial activity at the single cell level will provide radical new insights into the regulation of nitrogen fixation in chemosynthetic symbionts, shedding new light on the evolution of nitrogen-fixing symbioses in contrasting hosts and environments.
Copyright © 2020 Petersen and Yuen.
Figures

Similar articles
-
Chemosynthetic symbionts of marine invertebrate animals are capable of nitrogen fixation.Nat Microbiol. 2016 Oct 24;2(1):16195. doi: 10.1038/nmicrobiol.2016.195. Nat Microbiol. 2016. PMID: 27775707 Free PMC article.
-
Terrestrial-type nitrogen-fixing symbiosis between seagrass and a marine bacterium.Nature. 2021 Dec;600(7887):105-109. doi: 10.1038/s41586-021-04063-4. Epub 2021 Nov 3. Nature. 2021. PMID: 34732889 Free PMC article.
-
Nested interactions between chemosynthetic lucinid bivalves and seagrass promote ecosystem functioning in contaminated sediments.Front Plant Sci. 2022 Jul 22;13:918675. doi: 10.3389/fpls.2022.918675. eCollection 2022. Front Plant Sci. 2022. PMID: 35937361 Free PMC article.
-
Symbiotic diversity in marine animals: the art of harnessing chemosynthesis.Nat Rev Microbiol. 2008 Oct;6(10):725-40. doi: 10.1038/nrmicro1992. Nat Rev Microbiol. 2008. PMID: 18794911 Review.
-
Are legumes different? Origins and consequences of evolving nitrogen fixing symbioses.J Plant Physiol. 2022 Sep;276:153765. doi: 10.1016/j.jplph.2022.153765. Epub 2022 Jul 30. J Plant Physiol. 2022. PMID: 35952452 Review.
Cited by
-
Sulfur oxidation and reduction are coupled to nitrogen fixation in the roots of the salt marsh foundation plant Spartina alterniflora.Nat Commun. 2024 Apr 29;15(1):3607. doi: 10.1038/s41467-024-47646-1. Nat Commun. 2024. PMID: 38684658 Free PMC article.
-
Adaptations to nitrogen availability drive ecological divergence of chemosynthetic symbionts.PLoS Genet. 2024 May 31;20(5):e1011295. doi: 10.1371/journal.pgen.1011295. eCollection 2024 May. PLoS Genet. 2024. PMID: 38820540 Free PMC article.
-
Anaerobic Sulfur Oxidation Underlies Adaptation of a Chemosynthetic Symbiont to Oxic-Anoxic Interfaces.mSystems. 2021 Jun 29;6(3):e0118620. doi: 10.1128/mSystems.01186-20. Epub 2021 May 26. mSystems. 2021. PMID: 34058098 Free PMC article.
References
-
- Cavanaugh CM, McKiness ZP, Newton ILG, Stewart FJ. 2006. Marine chemosynthetic symbioses, p 475–507. In Dworkin M, Falkow SI, Rosenberg E, Schleifer K-H, Stackebrandt E (ed), The prokaryotes: an evolving electronic resource for the microbial community. Springer, New York, NY.
-
- Laurich JR, Dove R, Paillard C, Dufour SC. 2018. Life and death in facultative chemosymbioses: control of bacterial population dynamics in the Thyasiridae. Symbiosis 75:123–133. doi:10.1007/s13199-017-0525-0. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

