Intralacrimal Sustained Delivery of Rapamycin Shows Therapeutic Effects without Systemic Toxicity in a Mouse Model of Autoimmune Dacryoadenitis Characteristic of Sjögren's Syndrome
- PMID: 33356170
- PMCID: PMC8015671
- DOI: 10.1021/acs.biomac.0c01468
Intralacrimal Sustained Delivery of Rapamycin Shows Therapeutic Effects without Systemic Toxicity in a Mouse Model of Autoimmune Dacryoadenitis Characteristic of Sjögren's Syndrome
Abstract
Sjögren's syndrome (SS) is an autoimmune disease associated with severe exocrinopathy, which is characterized by profound lymphocytic infiltration (dacryoadenitis) and loss of function of the tear-producing lacrimal glands (LGs). Systemic administration of Rapamycin (Rapa) significantly reduces LG inflammation in the male Nonobese Diabetic (NOD) model of SS-associated autoimmune dacryoadenitis. However, the systemic toxicity of this potent immunosuppressant limits its application. As an alternative, this paper reports an intra-LG delivery method using a depot formulation comprised of a thermoresponsive elastin-like polypeptide (ELP) and FKBP, the cognate receptor for Rapa (5FV). Depot formation was confirmed in excised whole LG using cleared tissue and observation by both laser-scanning confocal and lightsheet microscopy. The LG depot was evaluated for safety, efficacy, and intra-LG pharmacokinetics in the NOD mouse disease model. Intra-LG injection with the depot formulation (5FV) retained Rapa in the LG for a mean residence time (MRT) of 75.6 h compared to Rapa delivery complexed with a soluble carrier control (5FA), which had a MRT of 11.7 h in the LG. Compared to systemic delivery of Rapa every other day for 2 weeks (seven doses), a single intra-LG depot of Rapa representing 16-fold less total drug was sufficient to inhibit LG inflammation and improve tear production. This treatment modality further reduced markers of hyperglycemia and hyperlipidemia while showing no evidence of necrosis or fibrosis in the LG. This approach represents a potential new therapy for SS-related autoimmune dacryoadenitis, which may be adapted for local delivery at other sites of inflammation; furthermore, these findings reveal the utility of optical imaging for monitoring the disposition of locally administered therapeutics.
Figures
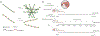
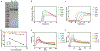


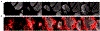




Similar articles
-
A rapamycin-binding protein polymer nanoparticle shows potent therapeutic activity in suppressing autoimmune dacryoadenitis in a mouse model of Sjögren's syndrome.J Control Release. 2013 Nov 10;171(3):269-79. doi: 10.1016/j.jconrel.2013.07.016. Epub 2013 Jul 25. J Control Release. 2013. PMID: 23892265 Free PMC article.
-
Berunda Polypeptides: Biheaded Rapamycin Carriers for Subcutaneous Treatment of Autoimmune Dry Eye Disease.Mol Pharm. 2019 Jul 1;16(7):3024-3039. doi: 10.1021/acs.molpharmaceut.9b00263. Epub 2019 May 30. Mol Pharm. 2019. PMID: 31095909 Free PMC article.
-
Rapamycin Eye Drops Suppress Lacrimal Gland Inflammation In a Murine Model of Sjögren's Syndrome.Invest Ophthalmol Vis Sci. 2017 Jan 1;58(1):372-385. doi: 10.1167/iovs.16-19159. Invest Ophthalmol Vis Sci. 2017. PMID: 28122086 Free PMC article.
-
Dysfunction of lacrimal and salivary glands in Sjögren's syndrome: nonimmunologic injury in preinflammatory phase and mouse model.J Biomed Biotechnol. 2011;2011:407031. doi: 10.1155/2011/407031. Epub 2011 Jun 1. J Biomed Biotechnol. 2011. PMID: 21660135 Free PMC article. Review.
-
Pathogenesis of Sjögren's syndrome: characteristics of different mouse models for autoimmune exocrinopathy.Clin Immunol. 2002 May;103(2):111-24. doi: 10.1006/clim.2002.5189. Clin Immunol. 2002. PMID: 12027416 Review. No abstract available.
Cited by
-
Hydra-Elastin-like Polypeptides Increase Rapamycin Potency When Targeting Cell Surface GRP78.Biomacromolecules. 2022 Aug 8;23(8):3116-3129. doi: 10.1021/acs.biomac.2c00048. Epub 2022 Jul 5. Biomacromolecules. 2022. PMID: 35786858 Free PMC article.
-
Local delivery strategies to restore immune homeostasis in the context of inflammation.Adv Drug Deliv Rev. 2021 Nov;178:113971. doi: 10.1016/j.addr.2021.113971. Epub 2021 Sep 13. Adv Drug Deliv Rev. 2021. PMID: 34530013 Free PMC article. Review.
-
Bet v 1-displaying elastin-like polypeptide nanoparticles induce a strong humoral and weak CD4+ T-cell response against Bet v 1 in a murine immunogenicity model.Front Immunol. 2022 Oct 6;13:1006776. doi: 10.3389/fimmu.2022.1006776. eCollection 2022. Front Immunol. 2022. PMID: 36275650 Free PMC article.
-
Protein and polypeptide mediated delivery to the eye.Adv Drug Deliv Rev. 2022 Sep;188:114441. doi: 10.1016/j.addr.2022.114441. Epub 2022 Jul 9. Adv Drug Deliv Rev. 2022. PMID: 35817213 Free PMC article. Review.
References
-
- Lemp MA, Dry eye (Keratoconjunctivitis Sicca), rheumatoid arthritis, and Sjogren’s syndrome. Am. J. Ophthalmol 2005, 140, (5), 898–9. - PubMed
-
- Nocturne G; Mariette X, Sjogren Syndrome-associated lymphomas: an update on pathogenesis and management. Br. J. Haematol 2015, 168, (3), 317–27. - PubMed
-
- Qin B; Wang J; Yang Z; Yang M; Ma N; Huang F; Zhong R, Epidemiology of primary Sjogren’s syndrome: a systematic review and meta-analysis. Ann. Rheum. Dis 2015, 74, (11), 1983–9. - PubMed
-
- Foulks GN; Forstot SL; Donshik PC; Forstot JZ; Goldstein MH; Lemp MA; Nelson JD; Nichols KK; Pflugfelder SC; Tanzer JM; Asbell P; Hammitt K; Jacobs DS, Clinical Guidelines for Management of Dry Eye Associated with Sjögren Disease. Ocul. Surf 2015, 13, (2), 118–132. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

