Effect of anti-IL17 and/or Rho-kinase inhibitor treatments on vascular remodeling induced by chronic allergic pulmonary inflammation
- PMID: 33357114
- PMCID: PMC7768836
- DOI: 10.1177/1753466620962665
Effect of anti-IL17 and/or Rho-kinase inhibitor treatments on vascular remodeling induced by chronic allergic pulmonary inflammation
Abstract
Background and aims: Expansion and morphological dysregulation of the bronchial vascular network occurs in asthmatic airways. Interleukin (IL) -17 and Rho-kinase (ROCK) are known to act in inflammation control and remodeling. Modulation of Rho-kinase proteins and IL-17 may be a promising approach for the treatment of asthma through the control of angiogenesis. Our objective was to analyze the effects of treatment with anti-IL17 and/or Rho-kinase inhibitor on vascular changes in mice with chronic allergic pulmonary inflammation.
Methods: Sixty-four BALB/c mice, with pulmonary inflammation induced by ovalbumin were treated with anti-IL17A (7.5/µg per dose, intraperitoneal) and/or Rho-kinase inhibitor (Y-27632-10 mg/kg, intranasal), 1 h before each ovalbumin challenge (22, 24, 26, and 28/days). Control animals were made to inhale saline. At the end of the protocol, lungs were removed, and morphometric analysis was performed to quantify vascular inflammatory, remodeling, and oxidative stress responses.
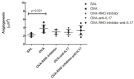
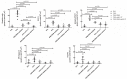
Results: Anti-IL17 or Rho-kinase inhibitor reduced the number of CD4+, CD8+, dendritic cells, IL-4, IL-5, IL-6, IL-10, IL-13, IL-17, Rho-kinase 1 and 2, transforming growth factor (TGF-β), vascular endothelial growth factor (VEGF), nuclear factor (NF)-KappaB, iNOS, metalloproteinase (MMP)-9, MMP-12, metalloproteinase inhibitor-1 (TIMP-1), FOXP-3, signal transducer and activator of transcription 1 (STAT1) and phospho-STAT1-positive cells, and actin, endothelin-1, isoprostane, biglycan, decorin, fibronectin and the collagen fibers volume fraction compared with the ovalbumin group (p < 0.05). The combination treatment, when compared with anti-IL17, resulted in potentiation of decrease in the number of IL1β- and dendritic cells-positive cells. When we compared the OVA-RHO inhibitor-anti-IL17 with OVA-RHO inhibitor we found a reduction in the number of CD8+ and IL-17, TGF-β, and phospho-STAT1-positive cells and endothelin-1 in the vessels (p < 0.05). There was an attenuation in the number of ROCK 2-positive cells in the group with the combined treatment when compared with anti-IL17 or Rho-kinase inhibitor-treated groups (p < 0.05).
Conclusion: We observed no difference in angiogenesis after treatment with Rho-kinase inhibitor and anti-IL17. Although the treatments did not show differences in angiogenesis, they showed differences in the markers involved in the angiogenesis process contributing to inflammation control and vascular remodeling.The reviews of this paper are available via the supplemental material section.
Keywords: Rho-kinase; Y-27632; angiogenesis; asthma; interleukin-17; neutralizing antibody; vascular remodeling.
Conflict of interest statement
Figures





References
-
- Barnig C, Frossard N, Levy BD. Towards targeting resolution pathways of airway inflammation in asthma. Pharmacol Ther 2018; 186: 98–113. - PubMed
-
- Xin L, Gao J, Ge X, et al. Increased pro-inflammatory cytokine-secreting regulatory T cells are correlated with the plasticity of T helper cell differentiation and reflect disease status in asthma. Respir Med 2018; 143: 129–138. - PubMed
-
- Lambrecht BN, Hammad H, Fahy JV. The cytokines of asthma. Immunity 2019; 50: 975–991. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

