Bortezomib, lenalidomide, and dexamethasone with or without elotuzumab in patients with untreated, high-risk multiple myeloma (SWOG-1211): primary analysis of a randomised, phase 2 trial
- PMID: 33357482
- PMCID: PMC8601389
- DOI: 10.1016/S2352-3026(20)30354-9
Bortezomib, lenalidomide, and dexamethasone with or without elotuzumab in patients with untreated, high-risk multiple myeloma (SWOG-1211): primary analysis of a randomised, phase 2 trial
Abstract
Background: The introduction of immunomodulatory agents, proteasome inhibitors, and autologous haematopoietic stem-cell transplantation has improved outcomes for patients with multiple myeloma, but patients with high-risk multiple myeloma have a poor long-term prognosis. We aimed to address optimal treatment for these patients.
Methods: SWOG-1211 is a randomised phase 2 trial comparing eight cycles of lenalidomide (25 mg orally on days 1-14 every 21 days), bortezomib (1·3 mg/m2 subcutaneously on days 1, 4, 8, and 11 every 21 days), and dexamethasone (20 mg orally on days 1, 2, 4, 5, 8, 9, 11, and 12 every 21 days; RVd) induction followed by dose-attenuated RVd maintenance (bortezomib 1 mg/m2 subcutaneously on days 1, 8, and 15; lenalidomide 15 mg orally on days 1-21; dexamethasone 12 mg orally on days 1, 18, and 15 every 28 days) until disease progression with or without elotuzumab (10 mg/kg intravenously on days 1, 8, and 15 for cycles 1-2, on days 1 and 11 for cycles 3-8, and on days 1 and 15 during maintenance). Patients were randomly assigned (1:1) to either RVd or RVd-elotuzumab. High-risk multiple myeloma was defined by one of the following: gene expression profiling high risk (GEPhi), t(14;16), t(14;20), del(17p) or amp1q21, primary plasma cell leukaemia and elevated serum lactate dehydrogenase (two times the upper limit of normal or more). The primary endpoint was progression-free survival, and all analyses were done on intention-to-treat basis among eligible patients who were evaluable for response. This study is registered with ClinicalTrials.gov, NCT01668719.
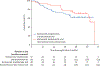
Findings: 100 (RVd n=52, RVd-elotuzumab n=48) patients were enrolled between Oct 27, 2013, and May 15, 2016, across 26 cooperative group institutions in the USA. Median age was 64 years (IQR 57-70, range 36-85). 74 (75%) of 99 had International Staging System stage II or stage III disease, 47 (47%) of 99 had amp1q21, 37 (37%) of 100 had del17p, 11 (11%) of 100 had t(14;16), eight (9%) of 90 were GEPhi, seven (7%) of 100 had primary plasma cell leukaemia, five (5%) of 100 had t(14;20), four (4%) of 100 had elevated serum lactate dehydrogenase, and 17 (17%) had two or more features. With a median follow-up of 53 months (IQR 46-59), no difference in median progression-free survival was observed (RVd 33·64 months [95% CI 19·55-not reached], RVd-elotuzumab 31·47 months [18·56-53·98]; hazard ratio 0·968 [80% CI 0·697-1·344]; one-sided p=0·45]. 37 (71%) of 52 patients in the RVd group and 37 (77%) of 48 in the RVd-elotuzumab group had grade 3 or worse adverse events. No significant differences in the safety profile were observed, although some notable results included grade 3-5 infections (four [8%] of 52 in the RVd group, eight [17%] of 48 in the RVd-elotuzumab group), sensory neuropathy (four [8%] of 52 in the RVd group, six [13%] of 48 in the RVd-elotuzumab group), and motor neuropathy (one [2%] of 52 in the RVd group, four [8%] of 48 in the RVd-elotuzumab group). There were no treatment-related deaths in the RVd group and one death in the RVd-elotuzumab group for which study treatment was listed as possibly contributing by the investigator.
Interpretation: In the first randomised study of high-risk multiple myeloma reported to date, the addition of elotuzumab to RVd induction and maintenance did not improve patient outcomes. However, progression-free survival in both study groups exceeded the original statistical assumptions and supports the role for continuous proteasome inhibitors and immunomodulatory drug combination maintenance therapy for this patient population.
Funding: National Institutes of Health, National Cancer Institute, Bristol Myers Squibb, Celgene, Leukemia and Lymphoma Society.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Figures
Comment in
-
High-risk myeloma: a challenge to define and to determine the optimal treatment.Lancet Haematol. 2021 Jan;8(1):e4-e6. doi: 10.1016/S2352-3026(20)30361-6. Epub 2020 Dec 22. Lancet Haematol. 2021. PMID: 33357481 No abstract available.
References
-
- Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014; 15: e538–48. - PubMed
-
- Usmani SZ, Rodriguez-Otero P, Bhutani M, Mateos MV, Miguel JS. Defining and treating high-risk multiple myeloma. Leukemia 2015; 29: 2119–25. - PubMed
-
- Fonseca R, Blood E, Rue M, et al. Clinical and biologic implications of recurrent genomic aberrations in myeloma. Blood 2003; 101: 4569–75. - PubMed
-
- Boersma-Vreugdenhil GR, Kuipers J, Van Stralen E, et al. The recurrent translocation t(14;20)(q32;q12) in multiple myeloma results in aberrant expression of MAFB: a molecular and genetic analysis of the chromosomal breakpoint. Br J Haematol 2004; 126: 355–63. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials