Sexual Dimorphism in Colon Cancer
- PMID: 33363037
- PMCID: PMC7759153
- DOI: 10.3389/fonc.2020.607909
Sexual Dimorphism in Colon Cancer
Abstract
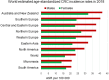
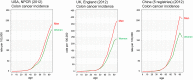
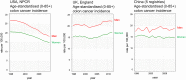
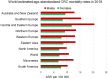
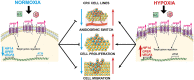
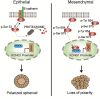
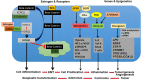
A higher incidence of colorectal cancer (CRC) is found in males compared to females. Young women (18-44 years) with CRC have a better survival outcome compared to men of the same age or compared to older women (over 50 years), indicating a global incidence of sexual dimorphism in CRC rates and survival. This suggests a protective role for the sex steroid hormone estrogen in CRC development. Key proliferative pathways in CRC tumorigenesis exhibit sexual dimorphism, which confer better survival in females through estrogen regulated genes and cell signaling. Estrogen regulates the activity of a class of Kv channels (KCNQ1:KCNE3), which control fundamental ion transport functions of the colon and epithelial mesenchymal transition through bi-directional interactions with the Wnt/β-catenin signalling pathway. Estrogen also modulates CRC proliferative responses in hypoxia via the novel membrane estrogen receptor GPER and HIF1A and VEGF signaling. Here we critically review recent clinical and molecular insights into sexual dimorphism of CRC biology modulated by the tumor microenvironment, estrogen, Wnt/β-catenin signalling, ion channels, and X-linked genes.
Keywords: GPER; Kv channels; Wnt/β-catenin; colon cancer; estrogen; sexual dimorphism.
Copyright © 2020 Abancens, Bustos, Harvey, McBryan and Harvey.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Sex Difference of Colon Adenoma Pathway and Colorectal Carcinogenesis.World J Mens Health. 2024 Apr;42(2):256-282. doi: 10.5534/wjmh.230085. Epub 2023 Aug 29. World J Mens Health. 2024. PMID: 37652658 Free PMC article. Review.
-
GPER Agonist G1 Prevents Wnt-Induced JUN Upregulation in HT29 Colorectal Cancer Cells.Int J Mol Sci. 2022 Oct 20;23(20):12581. doi: 10.3390/ijms232012581. Int J Mol Sci. 2022. PMID: 36293473 Free PMC article.
-
Bidirectional KCNQ1:β-catenin interaction drives colorectal cancer cell differentiation.Proc Natl Acad Sci U S A. 2017 Apr 18;114(16):4159-4164. doi: 10.1073/pnas.1702913114. Epub 2017 Apr 3. Proc Natl Acad Sci U S A. 2017. PMID: 28373572 Free PMC article.
-
A novel role for estrogen-induced signaling in the colorectal cancer gender bias.Ir J Med Sci. 2019 May;188(2):389-395. doi: 10.1007/s11845-018-1867-1. Epub 2018 Jul 17. Ir J Med Sci. 2019. PMID: 30014247 Review.
-
GPER mediates differential effects of estrogen on colon cancer cell proliferation and migration under normoxic and hypoxic conditions.Oncotarget. 2017 Sep 6;8(48):84258-84275. doi: 10.18632/oncotarget.20653. eCollection 2017 Oct 13. Oncotarget. 2017. PMID: 29137421 Free PMC article.
Cited by
-
Increased circulating regulatory T cells and decreased follicular T helper cells are associated with colorectal carcinogenesis.Front Immunol. 2024 Jan 26;15:1287632. doi: 10.3389/fimmu.2024.1287632. eCollection 2024. Front Immunol. 2024. PMID: 38343544 Free PMC article.
-
Therapeutic Strategies and Potential Actions of Female Sex Steroid Hormones and Their Receptors in Colon Cancer Based on Preclinical Studies.Life (Basel). 2022 Apr 18;12(4):605. doi: 10.3390/life12040605. Life (Basel). 2022. PMID: 35455096 Free PMC article. Review.
-
Sex Difference of Colon Adenoma Pathway and Colorectal Carcinogenesis.World J Mens Health. 2024 Apr;42(2):256-282. doi: 10.5534/wjmh.230085. Epub 2023 Aug 29. World J Mens Health. 2024. PMID: 37652658 Free PMC article. Review.
-
Intestinal microbiota regulates diabetes and cancer progression by IL-1β and NOX4 dependent signaling cascades.Cell Mol Life Sci. 2022 Sep;79(9):502. doi: 10.1007/s00018-022-04485-x. Epub 2022 Aug 30. Cell Mol Life Sci. 2022. PMID: 36040503 Free PMC article.
-
Sex disparities in outcomes among hospitalizations for heart failure.Sci Rep. 2024 Nov 7;14(1):27054. doi: 10.1038/s41598-024-77109-y. Sci Rep. 2024. PMID: 39511271 Free PMC article.
References
-
- Globocan Estimated cancer incidence, mortality and prevalence worldwide 2018. (2018). Available at: https://gco.iarc.fr/today/data/factsheets/cancers/10_8_9-Colorectum-fact...
-
- Cancer today Global Cancer Observatory. Available at: https://gco.iarc.fr/today/home.
Publication types
LinkOut - more resources
Full Text Sources

