NOD2/RIG-I Activating Inarigivir Adjuvant Enhances the Efficacy of BCG Vaccine Against Tuberculosis in Mice
- PMID: 33365029
- PMCID: PMC7751440
- DOI: 10.3389/fimmu.2020.592333
NOD2/RIG-I Activating Inarigivir Adjuvant Enhances the Efficacy of BCG Vaccine Against Tuberculosis in Mice
Abstract
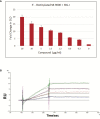
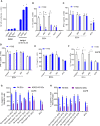
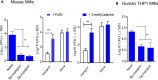
Tuberculosis (TB) caused by Mycobacterium tuberculosis (MTB) kills about 1.5 million people each year and the widely used Bacille Calmette-Guérin (BCG) vaccine provides a partial protection against TB in children and adults. Because BCG vaccine evades lysosomal fusion in antigen presenting cells (APCs), leading to an inefficient production of peptides and antigen presentation required to activate CD4 T cells, we sought to boost its efficacy using novel agonists of RIG-I and NOD2 as adjuvants. We recently reported that the dinucleotide SB 9200 (Inarigivir) derived from our small molecule nucleic acid hybrid (SMNH)® platform, activated RIG-I and NOD2 receptors and exhibited a broad-spectrum antiviral activity against hepatitis B and C, Norovirus, RSV, influenza and parainfluenza. Inarigivir increased the ability of BCG-infected mouse APCs to secrete elevated levels of IL-12, TNF-α, and IFN-β, and Caspase-1 dependent IL-1β cytokine. Inarigivir also increased the ability of macrophages to kill MTB in a Caspase-1-, and autophagy-dependent manner. Furthermore, Inarigivir led to a Capsase-1 and NOD2- dependent increase in the ability of BCG-infected APCs to present an Ag85B-p25 epitope to CD4 T cells in vitro. Consistent with an increase in immunogenicity of adjuvant treated APCs, the Inarigivir-BCG vaccine combination induced robust protection against tuberculosis in a mouse model of MTB infection, decreasing the lung burden of MTB by 1-log10 more than that afforded by BCG vaccine alone. The Inarigivir-BCG combination was also more efficacious than a muramyl-dipeptide-BCG vaccine combination against tuberculosis in mice, generating better memory T cell responses supporting its novel adjuvant potential for the BCG vaccine.
Keywords: BCG vaccine; NOD2; RIG-I; adjuvants; antigen presentation; inarigivir; inflammasome; tuberculosis.
Copyright © 2020 Khan, Singh, Mishra, Soudani, Bakhru, Singh, Zhang, Canaday, Sheri, Padmanabhan, Challa, Iyer and Jagannath.
Conflict of interest statement
RI is a shareholder of Spring Bank Pharmaceuticals, Inc. AS, SP, and SC own stock options in SBP. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

