Naringin attenuates renal interstitial fibrosis by regulating the TGF-β/Smad signaling pathway and inflammation
- PMID: 33365066
- PMCID: PMC7716641
- DOI: 10.3892/etm.2020.9498
Naringin attenuates renal interstitial fibrosis by regulating the TGF-β/Smad signaling pathway and inflammation
Abstract
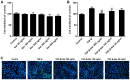
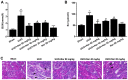
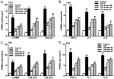
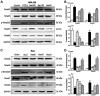
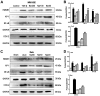
Interstitial fibrosis is a typical feature of all progressive renal diseases. The process of fibrosis is frequently coupled with the presence of pro-fibrotic factors and inflammation. Naringin is a dihydroflavone compound that has been previously reported to exhibit anti-fibrotic effects in the liver, where it prevents oxidative damage. In the present study, a rat model of renal interstitial fibrosis and fibrosis cell model were established to evaluate the effects of naringin on inflammatory proteins and fibrosis markers in kidney of rats and NRK-52E cells, and to elucidate the role of the TGF-β/Smad signaling pathway in this mechanism. Compared with those in fibrotic NRK-52E cells that were stimulated by transforming growth factor-β (TGF-β), gene expression levels of α-smooth muscle actin (α-SMA), collagen 1 (COL1A1), collagen 3 (COL3A1), interleukin (IL)-1β, IL-6 and tumor necrosis factor-α (TNF-α) were all found to be significantly decreased in fibrotic NRK-52E cells following treatment with naringin (50, 100 and 200 ng/ml). Results from the histopathological studies showed that naringin treatment preserved the renal tissue structure and reduced the degree of fibrosis in the kidney tissues of rats that underwent unilateral ureteral obstruction (UUO). In addition, naringin administration reduced the expression of α-SMA, COL1A1, COL3A1, IL-1β, IL-6 and TNF-α in the kidneys of rats following UUO. The current study, using western blot analysis, indicated that naringin also downregulated the activation of Smad2/3 and the expression of Smad4, high-mobility group protein B1, activator protein-1, NF-κB and cyclooxygenase-2 whilst upregulating the expression of Smad7 in fibrotic NRK-52E cells and rats in the UUO group. In conclusion, naringin could antagonize renal interstitial fibrosis by regulating the TGF-β/Smad pathway and the expression of inflammatory factors.
Keywords: anti-inflammatory; naringin; renal interstitial fibrosis; transforming growth factor-β/Smad.
Copyright: © Wang et al.
Figures



Similar articles
-
Anti-renal fibrosis effect of asperulosidic acid via TGF-β1/smad2/smad3 and NF-κB signaling pathways in a rat model of unilateral ureteral obstruction.Phytomedicine. 2019 Feb;53:274-285. doi: 10.1016/j.phymed.2018.09.009. Epub 2018 Sep 5. Phytomedicine. 2019. PMID: 30668407
-
[Mechanisms of cordycepin on improving renal interstitial fibrosis via regulating eIF2α/TGF-β/Smad signaling pathway].Zhongguo Zhong Yao Za Zhi. 2014 Nov;39(21):4096-101. Zhongguo Zhong Yao Za Zhi. 2014. PMID: 25775775 Chinese.
-
Carvacrol preserves antioxidant status and attenuates kidney fibrosis via modulation of TGF-β1/Smad signaling and inflammation.Food Funct. 2022 Oct 17;13(20):10587-10600. doi: 10.1039/d2fo01384c. Food Funct. 2022. PMID: 36156620
-
Central role of dysregulation of TGF-β/Smad in CKD progression and potential targets of its treatment.Biomed Pharmacother. 2018 May;101:670-681. doi: 10.1016/j.biopha.2018.02.090. Epub 2018 Mar 22. Biomed Pharmacother. 2018. PMID: 29518614 Review.
-
Transforming growth factor-beta and Smad signalling in kidney diseases.Nephrology (Carlton). 2005 Feb;10(1):48-56. doi: 10.1111/j.1440-1797.2005.00334.x. Nephrology (Carlton). 2005. PMID: 15705182 Review.
Cited by
-
DACH1 attenuated PA-induced renal tubular injury through TLR4/MyD88/NF-κB and TGF-β/Smad signalling pathway.J Endocrinol Invest. 2024 Jun;47(6):1531-1544. doi: 10.1007/s40618-023-02253-7. Epub 2023 Dec 26. J Endocrinol Invest. 2024. PMID: 38147289
-
Dietary naringin supplementation on laying performance and antioxidant capacity of Three-Yellow breeder hens during the late laying period.Poult Sci. 2022 Sep;101(9):102023. doi: 10.1016/j.psj.2022.102023. Epub 2022 Jun 23. Poult Sci. 2022. PMID: 35901650 Free PMC article.
-
Regulatory mechanism and therapeutic potentials of naringin against inflammatory disorders.Heliyon. 2024 Jan 18;10(3):e24619. doi: 10.1016/j.heliyon.2024.e24619. eCollection 2024 Feb 15. Heliyon. 2024. PMID: 38317884 Free PMC article. Review.
-
Recombinant TSG-6 protein inhibits the growth of capsule fibroblasts in frozen shoulder via suppressing the TGF-β/Smad2 signal pathway.J Orthop Surg Res. 2021 Sep 15;16(1):564. doi: 10.1186/s13018-021-02705-x. J Orthop Surg Res. 2021. PMID: 34526039 Free PMC article.
-
Unraveling TGF-β1's Role in Mediating Fibrosis and Cell Death in Feline Kidney Cells.Animals (Basel). 2025 Jan 17;15(2):257. doi: 10.3390/ani15020257. Animals (Basel). 2025. PMID: 39858257 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
