Mitochondrial Transfer and Regulators of Mesenchymal Stromal Cell Function and Therapeutic Efficacy
- PMID: 33365311
- PMCID: PMC7750467
- DOI: 10.3389/fcell.2020.603292
Mitochondrial Transfer and Regulators of Mesenchymal Stromal Cell Function and Therapeutic Efficacy
Abstract
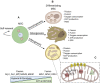
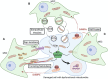
Mesenchymal stromal cell (MSC) metabolism plays a crucial role in the surrounding microenvironment in both normal physiology and pathological conditions. While MSCs predominantly utilize glycolysis in their native hypoxic niche within the bone marrow, new evidence reveals the importance of upregulation in mitochondrial activity in MSC function and differentiation. Mitochondria and mitochondrial regulators such as sirtuins play key roles in MSC homeostasis and differentiation into mature lineages of the bone and hematopoietic niche, including osteoblasts and adipocytes. The metabolic state of MSCs represents a fine balance between the intrinsic needs of the cellular state and constraints imposed by extrinsic conditions. In the context of injury and inflammation, MSCs respond to reactive oxygen species (ROS) and damage-associated molecular patterns (DAMPs), such as damaged mitochondria and mitochondrial products, by donation of their mitochondria to injured cells. Through intercellular mitochondria trafficking, modulation of ROS, and modification of nutrient utilization, endogenous MSCs and MSC therapies are believed to exert protective effects by regulation of cellular metabolism in injured tissues. Similarly, these same mechanisms can be hijacked in malignancy whereby transfer of mitochondria and/or mitochondrial DNA (mtDNA) to cancer cells increases mitochondrial content and enhances oxidative phosphorylation (OXPHOS) to favor proliferation and invasion. The role of MSCs in tumor initiation, growth, and resistance to treatment is debated, but their ability to modify cancer cell metabolism and the metabolic environment suggests that MSCs are centrally poised to alter malignancy. In this review, we describe emerging evidence for adaptations in MSC bioenergetics that orchestrate developmental fate decisions and contribute to cancer progression. We discuss evidence and potential strategies for therapeutic targeting of MSC mitochondria in regenerative medicine and tissue repair. Lastly, we highlight recent progress in understanding the contribution of MSCs to metabolic reprogramming of malignancies and how these alterations can promote immunosuppression and chemoresistance. Better understanding the role of metabolic reprogramming by MSCs in tissue repair and cancer progression promises to broaden treatment options in regenerative medicine and clinical oncology.
Keywords: MSC differentiation; cancer metabolism; hematological malignancy; mesenchymal stromal cells (MSCs); metabolic reprogramming; mitochondrial biogenesis; mitochondrial dynamics; mitochondrial transfer.
Copyright © 2020 Mohammadalipour, Dumbali and Wenzel.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Adekola K. U. A., Dalva Aydemir S., Ma S., Zhou Z., Rosen S. T., Shanmugam M. (2015). Investigating and targeting chronic lymphocytic leukemia metabolism with the human immunodeficiency virus protease inhibitor ritonavir and metformin. Leuk. Lymphoma 56, 450–459. 10.3109/10428194.2014.922180 - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

