Diesel Exhaust Particulates Induce Neutrophilic Lung Inflammation by Modulating Endoplasmic Reticulum Stress-Mediated CXCL1/KC Expression in Alveolar Macrophages
- PMID: 33371364
- PMCID: PMC7767360
- DOI: 10.3390/molecules25246046
Diesel Exhaust Particulates Induce Neutrophilic Lung Inflammation by Modulating Endoplasmic Reticulum Stress-Mediated CXCL1/KC Expression in Alveolar Macrophages
Abstract
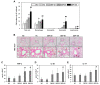
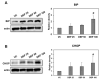
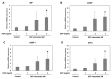
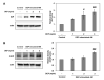
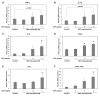
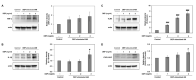
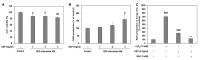
Diesel exhaust particulates (DEP) have adverse effects on the respiratory system. Endoplasmic reticulum (ER) abnormalities contribute to lung inflammation. However, the relationship between DEP exposure and ER stress in the respiratory immune system and especially the alveolar macrophages (AM) is poorly understood. Here, we examined ER stress and inflammatory responses using both in vivo and in vitro study. For in vivo study, mice were intratracheally instilled with 25, 50, and 100 μg DEP and in vitro AM were stimulated with DEP at 1, 2, and 3 mg/mL. DEP increased lung weight and the number of inflammatory cells, especially neutrophils, and inflammatory cytokines in bronchoalveolar lavage fluid of mice. DEP also increased the number of DEP-pigmented AM and ER stress markers including bound immunoglobulin protein (BiP) and CCAAT/enhancer binding protein-homologous protein (CHOP) were upregulated in the lungs of DEP-treated mice. In an in vitro study, DEP caused cell damage, increased intracellular reactive oxygen species, and upregulated inflammatory genes and ER stress-related BiP, CHOP, splicing X-box binding protein 1, and activating transcription factor 4 expressions in AM. Furthermore, DEP released the C-X-C Motif Chemokine Ligand 1 (CXCL1/KC) in AM. In conclusion, DEP may contribute to neutrophilic lung inflammation pathogenesis by modulating ER stress-mediated CXCL1/KC expression in AM.
Keywords: alveolar macrophages; chemokine CXCL1/KC; diesel exhaust particulate; endoplasmic reticulum stress; neutrophilic lung inflammation; particulate matter 2.5.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
TF-343 Alleviates Diesel Exhaust Particulate-Induced Lung Inflammation via Modulation of Nuclear Factor-κB Signaling.J Immunol Res. 2019 Oct 30;2019:8315845. doi: 10.1155/2019/8315845. eCollection 2019. J Immunol Res. 2019. PMID: 31781683 Free PMC article.
-
Physiological and inflammatory responses in an anthropomorphically relevant model of acute diesel exhaust particle exposure are sex and dose-dependent.Inhal Toxicol. 2011 Dec;23(14):906-17. doi: 10.3109/08958378.2011.625454. Inhal Toxicol. 2011. PMID: 22122304
-
Lung inflammation and genotoxicity in mice lungs after pulmonary exposure to candle light combustion particles.Toxicol Lett. 2017 Jul 5;276:31-38. doi: 10.1016/j.toxlet.2017.04.015. Epub 2017 Apr 29. Toxicol Lett. 2017. PMID: 28465192
-
Diesel Exhaust Particles and the Induction of Macrophage Activation and Dysfunction.Inflammation. 2018 Feb;41(1):356-363. doi: 10.1007/s10753-017-0682-6. Inflammation. 2018. PMID: 29047037 Review.
-
The dual effect of the particulate and organic components of diesel exhaust particles on the alteration of pulmonary immune/inflammatory responses and metabolic enzymes.J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2002 Nov;20(2):117-47. doi: 10.1081/GNC-120016202. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2002. PMID: 12515672 Review.
Cited by
-
Age and Gender Effects on Genotoxicity in Diesel Exhaust Particles Exposed C57BL/6 Mice.Biomolecules. 2021 Mar 2;11(3):374. doi: 10.3390/biom11030374. Biomolecules. 2021. PMID: 33801497 Free PMC article.
-
Diagnostic model constructed by five EMT-related genes for renal fibrosis and reflecting the condition of immune-related cells.Front Immunol. 2023 May 17;14:1161436. doi: 10.3389/fimmu.2023.1161436. eCollection 2023. Front Immunol. 2023. PMID: 37266443 Free PMC article.
-
Establishment of an artificial particulate matter-induced lung disease model through analyzing pathological changes and transcriptomic profiles in mice.Sci Rep. 2023 Apr 12;13(1):5955. doi: 10.1038/s41598-023-29919-9. Sci Rep. 2023. PMID: 37045933 Free PMC article.
-
Diesel Exhaust Particulates Enhances Susceptibility of LPS-Induced Acute Lung Injury through Upregulation of the IL-17 Cytokine-Derived TGF-β1/Collagen I Expression and Activation of NLRP3 Inflammasome Signaling in Mice.Biomolecules. 2021 Jan 6;11(1):67. doi: 10.3390/biom11010067. Biomolecules. 2021. PMID: 33419073 Free PMC article.
-
Coconut Oil Alleviates the Oxidative Stress-Mediated Inflammatory Response via Regulating the MAPK Pathway in Particulate Matter-Stimulated Alveolar Macrophages.Molecules. 2022 May 2;27(9):2898. doi: 10.3390/molecules27092898. Molecules. 2022. PMID: 35566249 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

