Interface between Bats and Pigs in Heavy Pig Production
- PMID: 33375071
- PMCID: PMC7822039
- DOI: 10.3390/v13010004
Interface between Bats and Pigs in Heavy Pig Production
Abstract
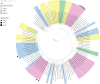
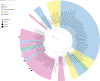
Bats are often claimed to be a major source for future viral epidemics, as they are associated with several viruses with zoonotic potential. Here we describe the presence and biodiversity of bats associated with intensive pig farms devoted to the production of heavy pigs in northern Italy. Since chiropters or signs of their presence were not found within animal shelters in our study area, we suggest that fecal viruses with high environmental resistance have the highest likelihood for spillover through indirect transmission. In turn, we investigated the circulation of mammalian orthoreoviruses (MRVs), coronaviruses (CoVs) and astroviruses (AstVs) in pigs and bats sharing the same environment. Results of our preliminary study did not show any bat virus in pigs suggesting that spillover from these animals is rare. However, several AstVs, CoVs and MRVs circulated undetected in pigs. Among those, one MRV was a reassortant strain carrying viral genes likely acquired from bats. On the other hand, we found a swine AstV and a MRV strain carrying swine genes in bat guano, indicating that viral exchange at the bat-pig interface might occur more frequently from pigs to bats rather than the other way around. Considering the indoor farming system as the most common system in the European Union (EU), preventive measures should focus on biosecurity rather than displacement of bats, which are protected throughout the EU and provide critical ecosystem services for rural settings.
Keywords: bats; pigs; reassortment; spillover; viruses.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


References
-
- Plowright R.K., Foley P., Field H.E., Dobson A.P., Foley J.E., Eby P., Daszak P. Urban habituation, ecological connectivity and epidemic dampening: The emergence of Hendra virus from flying foxes (Pteropus spp.) Proc. R. Soc. B Biol. Sci. 2011;278:3703–3712. doi: 10.1098/rspb.2011.0522. - DOI - PMC - PubMed
-
- Rulli M.C., D’Odorico P., Galli N., Hayman D.T.S. Land Use Change and Coronavirus Emergence Risk. medRxiv. 2020 doi: 10.1101/2020.07.31.20166090. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

