Similar Repair Effects of Human Placenta, Bone Marrow Mesenchymal Stem Cells, and Their Exosomes for Damaged SVOG Ovarian Granulosa Cells
- PMID: 33376492
- PMCID: PMC7738794
- DOI: 10.1155/2020/8861557
Similar Repair Effects of Human Placenta, Bone Marrow Mesenchymal Stem Cells, and Their Exosomes for Damaged SVOG Ovarian Granulosa Cells
Abstract
Background: This study is aimed at investigating the repairing effect of mesenchymal stem cells and their exosomes from different sources on ovarian granulosa cells damaged by chemotherapy drugs-phosphoramide mustard (PM).
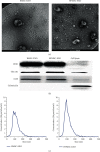
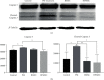
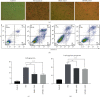
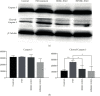
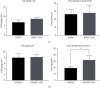
Methods: In this study, we choose bone marrow mesenchymal stem cells (BMSCs) and human placental mesenchymal stem cells (HPMSCs) for research. Then, they were cocultured with human ovarian granulosa cells (SVOG) injured by phosphoramide mustard (PM), respectively. β-Galactosidase staining, flow cytometry, and Western blot were used to detect the changes in the senescence and apoptosis of SVOG cells before and after their coculture with the above two types of MSCs. Subsequently, exosomes from these two types of MSCs were extracted and added to the culture medium of SVOG cells after PM injury to test whether these two types of exosomes played a role similar to that of MSCs in repairing damaged SVOG cells.
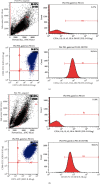
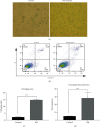
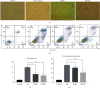
Results: PM treatment-induced apoptotic SVOG cells were significantly decreased after HPMSCs and BMSCs as compared with control group. After coculturing with these two types of MSCs, PM-treated SVOG cells showed significantly reduced senescence and apoptosis proportions as well as cleaved-Caspase 3 expression, and HPMSCs played a slightly stronger role than BMSCs in repairing SVOG cells in terms of the above three indicators. In addition, the ratios of senescent and apoptotic SVOG cells were also significantly reduced by the two types of exosomes, which played a role similar to that of MSCs in repairing cell damages.
Conclusions: The results indicated that BMSCs, HPMSCs, and their exosomes all exerted a certain repair effect on SVOG cells damaged by PM, and consistent repair effect was observed between exosomes and MSCs. The repair effect of exosomes secreted from BMSCs and HPMSCs on the SVOG cells was studied for the first time, and the results fully demonstrated that exosomes are the key carriers for MSCs to play their role.
Copyright © 2020 Shuwen Chen et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Tilmann C., Capel B. Mesonephric cell migration induces testis cord formation and Sertoli cell differentiation in the mammalian gonad. Development. 1999;126(13):2883–2890. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

